The solubility of phenol in water at 25 C is 87 g/L. The solubility of naphthol at
Question:
The solubility of phenol in water at 25 °C is 87 g/L. The solubility of naphthol at the same temperature is only 0.74 g/L. Examine the structures of phenol and naphthol shown here and explain why phenol is so much more soluble than naphthol.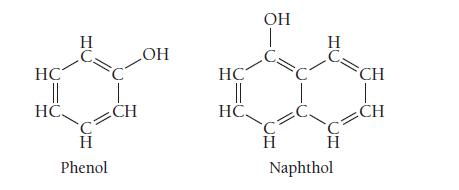
Transcribed Image Text:
HC || HC Н C. Н Phenol I CH OH HC || HC. OH н H Н Naphthol CH CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Phenol is more soluble in water than naphthol because it is more polar The hydroxyl group OH in phen...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following table presents the solubilities of several gases in water at 25 C under a total pressure of gas and water vapor of 1 atm. (a) What volume of CH 4 (g) under standard conditions of...
-
The solubility of carbon tetrachloride (CCl 4 ) in water at 25 C is 1.2 g/L. The solubility of chloroform (CHCl 3 ) at the same temperature is 10.1 g/L. Why is chloroform almost ten times more...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Why is it that I got 0 Row with nothing showing up? Is there a flow in my logic? Using AdventureWorks DW2017, list all the Canadian customers who have spent more than $5,000 total. Show Customerkey,...
-
Identify several types of printers. What are the advantages and disadvantages of each type?
-
The critical resolved shear stress for iron is 27MPa (4000psi). Determine the maximum possible yield strength for a single crystal of Fe pulled in tension.
-
Go to the St. Louis Federal Reserve FRED database, and find data on real GDP (GDPC1), potential GDP (GDPPOT), and the unemployment rate (UNRATE) from 1960 to the most recent period. For the...
-
Presented below are the captions of Nikos Companys balance sheet. (a) Current assets. (b) Investments. (c) Property, plant, and equipment. (d) Intangible assets . (e) Other assets. (f) Current...
-
Problem 4. Consider the following imperfect information game. Notice that both play- ers have two information sets. 5,2 1 a b 2 t 1 d d 4,3 6.0 4,6 0,4 6,0 (a) Find all subgame perfect Nash...
-
Potassium perchlorate (KClO 4 ) has a lattice energy of -599 kJ/mol and a heat of hydration of -548 kJ/mol. Find the heat of solution for potassium perchlorate and determine the temperature change...
-
An aqueous CaCl 2 solution has a vapor pressure of 81.6 mmHg at 50 C. The vapor pressure of pure water at this temperature is 92.6 mmHg. What is the concentration of CaCl 2 in mass percent?
-
During 2018, Fueltronics spends $200,000 on several different research projects to gain new knowledge about the use of alternative fuels to operate motorized vehicles. Because one of the projects...
-
If youre not familiar with vesting, the idea is that when a firm is launched, instead of issuing stock outright to the founders, the stock is distributed over a period of time, typically three to...
-
Briefly describe each of the five stages in the organizational life cycle.
-
Kelly Andrews has developed a new line of jewelry that has created some positive buzz among friends and some business stores in her local community. When asked by a reporter, Where do you plan to...
-
Spend some time studying PurBlu Beverages, the subject of the studentinitiated business profiled at the beginning of the chapter. In what ways have PurBlu Beverages business practices and its...
-
ModCloth founders Susan Koger and Eric Koger met in high school. Susan enjoyed shopping for vintage clothing, and over time accumulated so many outfits that she decided to sell some. Eric, who was...
-
Consider the most appropriate policy for tackling each of the different types of unemployment.
-
State whether each of the following will increase or decrease the power of a one-way between-subjects ANOVA. (a) The effect size increases. (b) Mean square error decreases. (c) Mean square between...
-
Making use of the expression I(y) = I 0 e -y [4.78] for an absorbing medium, we define a quantity called the unit transmittance T 1 . At normal incidence, Eq. (4.55), T = I t /I i , and thus when y =...
-
Show that at normal incidence on the boundary between two dielectrics, as n ti S 1, R 0, and T 1. Moreover, prove that as n ti 1, R 0, and T 1 Moreover, prove that as n ti 1, R || 0, R 0, T...
-
Derive the expressions for r ¥ and r || given by Eqs. (4.70) and (4.71). cos 6; (n sin 0;)'/ cos 0; + (n sin 0;)'/2 (4.70) ni cos 0; (ni sin e,)'/2 n cos e; + (ni sin0;}'/2 and (4.71)
-
Show that the density of a 5kg solid cylinder that is 10cm tall with a radius of 3cm is 17.7g/cm3. V=rh 3cm 10cm
-
2. A col encloses an area of 5.0 10-4 m. The plane of the coil is positioned at 60 to the direction of a magnetic field of 0.25 T. B 60 B 8x What is the value of the magnetic flux through the coil?...
-
A woman at an airport is towing her 14.0 kg suitcase at constant speed by pulling on a strap at an angle 0 above the horizontal (see figure). She pulls on the strap with a 28.5 N force, and the...

Study smarter with the SolutionInn App


