The tabulated data show the rate constant of a reaction measured at several different temperatures. Use an
Question:
The tabulated data show the rate constant of a reaction measured at several different temperatures. Use an Arrhenius plot to determine the activation barrier and frequency factor for the reaction.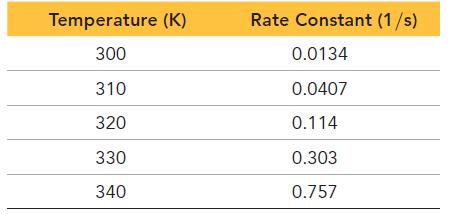
Transcribed Image Text:
Temperature (K) 300 310 320 330 340 Rate Constant (1/s) 0.0134 0.0407 0.114 0.303 0.757
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To determine the activation barrier and frequency factor for the reaction we ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The tabulated data show the rate constant of a reaction measured at several different temperatures. Use an Arrhenius plot to determine the activation barrier and frequency factor for the reaction....
-
The following data show the rate constant of a reaction measuredat several different temperatures. Temperature (K) Rate Constant (1/s) 300 7.5610 - 2 310 0.221 320 0.605 330 1.56 340 3.79 Part A Use...
-
The data shown here were collected for the first-order reaction: Use an Arrhenius plot to determine the activation barrier and frequency factor for the reaction. NO(g) N(g) + O(g)
-
What is unrealistic about the domino fault model?
-
Personal Electronix sells iPads and iPods. The business is divided into two divisions along product lines. CVP income statements for a recent quarter's activity are presented below. Instructions (a)...
-
Answer the following questions related to Dubois Inc. (a) Dubois Inc. has $600,000 to invest. The company is trying to decide between two alternative uses of the funds. One alternative provides...
-
Apply the marketing-myopia concept to print media, magazines, and newspapers. What is the implication?
-
You are provided with the following information for Koetteritz Inc. for the month ended June 30, 2017. Koetteritz uses the periodic method for inventory. Instructions (a) Calculate (i) Ending...
-
12. Your calculations show that if the wrong link is shared to a potential Democratic voter, there is a strong chance it could result in them changing their vote to Independent or Republican. If you...
-
The tabulated data were collected for the second-order reaction: Cl(g) + H 2 (g) HCl(g) + H(g) Use an Arrhenius plot to determine the activation barrier and frequency factor for the reaction....
-
The rate constant (k) for a reaction was measured as a function of temperature. A plot of ln k versus 1/T (in K) is linear and has a slope of -1.01 * 10 4 K. Calculate the activation energy for the...
-
Bath Co. is a company specializing in the manufacture and sale of baths. Each bath consists of a main unit plus a set of bath fittings. The company is split into two divisions, A and B. Division A...
-
In May Joseph Laval was appointed managing director of the FrenchThai joint venture mobile telephony company Siammobile. The personnel in the joint venture were people who had transferred from the...
-
How can multilateral funds improve the support of a green and resilient development policy in emerging economies?
-
Electric vehicle (EV) reliability problems are frequently reported by drivers; however, for the first time, the rate of EV problems is lower than the rate of non-EV problems. Are urban areas prepared...
-
Due to climate change it is necessary to improve research skills and firm-level innovation in the agriculture sector. Discuss the experience that occurred in Carmens internship.
-
How much control should the HR department possess in a business/company?
-
On January 1, 2013, Wells Corp. sold $100,000 of its own 6 percent, 10-year bonds. Interest ispayable annually on December 31. The bonds were sold to yield an effective interest rate of 5percent....
-
Use a calculator to evaluate the expression. Round your result to the nearest thousandth. V (32 + #)
-
Given the circuit in Fig. 16.65 , find i(t) and v(t) for t > 0. i(t) 1H v(t) 1 2 t = 0 180 V (+ 1/4 ll
-
Find the output voltage v o (t) in the circuit of Fig. 16.64 . t= 0 10 1H3 10 mF 1.5 A () 52 Vo
-
In the circuit of Fig. 16.63 , find v(t) and i(t) for t > 0. Assume v(0) = 0 V and i(0) = 1.25 A. 5u(t) A + 0.5 F 1H ll
-
Define x = 0 and x+1 = x -[(tan x-1)/sec x,]. What is lim, x, in this example? Relate this to Newton's method.
-
6. Student Survey: In a recent survey, the following data were obtained in response to the question, "If the number of summer classes were increased, would you be more likely to enroll in one or more...
-
How many roots does the equation z4 6z + 3 = 0 have in the disk |z| < 2? and in |z| < 1?

Study smarter with the SolutionInn App


