This table displays the vapor pressure of ammonia at several different temperatures. Use the data to determine
Question:
This table displays the vapor pressure of ammonia at several different temperatures. Use the data to determine the heat of vaporization and normal boiling point of ammonia.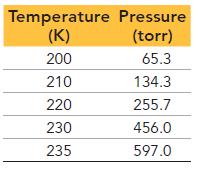
Transcribed Image Text:
Temperature Pressure (K) (torr) 200 65.3 210 134.3 220 255.7 230 456.0 235 597.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Hvap ...View the full answer

Answered By

Michael Owens
I am a competent Software Engineer with sufficient experience in web applications development using the following programming languages:-
HTML5, CSS3, PHP, JAVASCRIPT, TYPESCRIPT AND SQL.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
This table displays the vapor pressure of nitrogen at several different temperatures. Use the data to determine the heat of vaporization and normal boiling point of nitrogen. Temperature Pressure (K)...
-
a) Sputtering is a very important physical process in plasma engineering of thin films and coatings. In one or two paragraphs describe what sputtering is. Illustrate your answer with diagrams and...
-
Procurement of commodities is a critical activity for a company's survival, particularly in the manufacturing industry. It is essential in the industry because it standardizes the procurement of...
-
TABLE 6A-1 General Depreciation System: 200% or 150% Declining Balance Switching to Straight-Line* Half-Year Convention Recovery Year 3-Year 5-Year 7-Year 10-Year 15-Year 20-Year 1 33.33 20.00 14.29...
-
The Public Service Commission of State X issued a regulation completely banning all advertising that promotes the use of electricity by any electric utility company in State X. The commission issued...
-
Use source transformation to find Io in the circuit in figure. ww 4 kn 18 V 36 kn 4 kn 6 kn 6 kn 2 mA( 3 1 mA
-
Use the financial data for Randa Merchandising, Inc., in Exercise 13-13 to prepare its income statement for calendar-year 2017. (Ignore the earnings per share section.) Data From Exercise 13.13 In...
-
The Pitts Barbecue Company makes three kinds of barbecue sauce: Extra Hot, Hot, and Mild. Pitts vice president of marketing estimates that the company can sell 8,000 cases of its Extra Hot sauce plus...
-
The schedule below produces same outcome as the serial schedule < T1, T5>. But it is not view serializable. Why? T read (A) A := A-50 write (A) read (B) B := B+50 write (B) TS read (B) B: B-10 write...
-
Ethanol has a heat of vaporization of 38.56 kJ/mol and a normal boiling point of 78.4 C. What is the vapor pressure of ethanol at 15 C?
-
Suppose that 1.15 g of rubbing alcohol (C 3 H 8 O) evaporates from a 65.0-g aluminum block. If the aluminum block is initially at 25 C, what is the final temperature of the block after the...
-
Suppose 30% of the cells are young. Consider again the cells in Section 6.4, Exercises 29 and 30, but suppose that older cells, instead of not staining as often, do not stain as well. In particular,...
-
In order for a student to go on the road with the band, they need to return a permission slip signed by their parent or guardian. In cell C12, enter a VLOOKUP to retrieve the value from the...
-
Mary is an accountant and having an annual income of $400,000. She plans to open a bookstore and quitting her job. She has her own shop and the annual rent is $148,000. She expects the annual revenue...
-
Your department has a total salary budget of $62,500. Among your employees, there is a manager who is allocated a significant portion of this budget. 4. Calculate the manager's salary, given that it...
-
Talal Properties need a construction machinery costing AED 45,000. The machine will work for 5 years. Expected net cash inflows [in AED] are y1: 30,000; y2: 25,000; y3 :15,000; y4: 10,000 and y5:...
-
Hill Corporation issued $3,000,000 of 7% bonds at 98 on January 2, 2019. Interest is paid semiannually on June 30 and December 31. The bonds had a 10-year life from the date of issue, and the company...
-
Refer to exercise 9-24. Kurt, the manager of the Ohio unit, is unhappy with the results of the controller's study. He asks the controller to develop separate rates for fixed and variable costs in the...
-
Which task is performed by a book-keeper? A. Analysing the trading results B. Entering transactions in the ledger C. Preparing year-end financial statements D. Providing information for...
-
Predict the major product(s) of the reaction between l-valine and: (a) MeOH, H + (b) Di-tert-butyl-dicarbonate (c) NaOH, H 2 O (d) HCl
-
When the N terminus of a peptide is acetylated, the peptide derivative that is formed is unreactive toward phenyl isothiocyanate. Explain. Acetylation PEPTIDE PEPTIDE . N. N. Z
-
Glucagon is a peptide hormone produced by the pancreas that, with insulin, regulates blood glucose levels. Glucagon is comprised of 29 amino acid residues. Treatment with trypsin yields four...
-
A charged rod extends along the y axis from -2.00 to +2.00 m. The charge distribution on the rod is given by the linear charge density function = 10.0 lyl, all units Sl. The point P is (3.00, 0) m....
-
Consider the diver shown in the figure diving from a 10-m high platform. The diver completes 1.5 rotations, while also rising and falling vertically and moving forward horizontally. We will assume...
-
Portability is the ability of software to move easily from one hardware platform to another. UNIX was the first portable operating system, allowing programs to compile on multiple hardware platforms...

Study smarter with the SolutionInn App


