This table displays the vapor pressure of nitrogen at several different temperatures. Use the data to determine
Question:
This table displays the vapor pressure of nitrogen at several different temperatures. Use the data to determine the heat of vaporization and normal boiling point of nitrogen.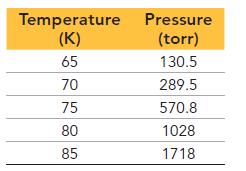
Transcribed Image Text:
Temperature Pressure (K) (torr) 65 130.5 289.5 570.8 1028 1718 70 75 80 85
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To determine the heat of vaporization and normal boiling point of nitrogen we can use the ClausiusCl...View the full answer

Answered By

Shristi Singh
A freshman year metallurgy and material science student in India.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
This table displays the vapor pressure of ammonia at several different temperatures. Use the data to determine the heat of vaporization and normal boiling point of ammonia. Temperature Pressure (K)...
-
Procurement of commodities is a critical activity for a company's survival, particularly in the manufacturing industry. It is essential in the industry because it standardizes the procurement of...
-
Suppose the vapor pressure of a substance is measured at two different temperatures. (a) By using the Clausius-Clapeyron equation (Equation 11.1) derive the following relationship between the vapor...
-
The management of a New York area investment firm wants to find out about the investment needs of its existing customers, for which it has an extensively detailed list, as a function of their...
-
EZRest Motel is a motel with 216 rooms located in the center of a large city in State Y. It is readily accessible from two interstate highways and three major State highways. The motel solicits...
-
Using source transformation, find Io in the circuit in figure. 8 kn 2 kn 2 mA 1 mA ww 3 kn 6 kn3
-
Use the same facts as problem 20 and prepare entries using the following independent assumptions: a. Lease A is abandoned in 2022. b. Lease A is proved in 2022. c. Insignificant Lease Y, with a cost...
-
Assume that Olive Corporation, in Comprehensive Problem 1, is an S corporation owned 50 percent by Linda Holiday and 50 percent by Ralph Winston. The corporation is not subject to any special taxes....
-
Question 3.1 Create a class containing a method name createArray and a main method. The method createArray creates an array where each element contains the square of its index. The size of the array...
-
Ethanol has a heat of vaporization of 38.56 kJ/mol and a normal boiling point of 78.4 C. What is the vapor pressure of ethanol at 15 C?
-
Suppose that 1.15 g of rubbing alcohol (C 3 H 8 O) evaporates from a 65.0-g aluminum block. If the aluminum block is initially at 25 C, what is the final temperature of the block after the...
-
If a firm's total revenue is $5 billion, its fixed costs are $3 billion, and its variable cost are $1.5 billion, what does it do for a) In the short run? b) In the long run?
-
Consider: BSL Racket has a function named modulo, that can be described as follows: Assume you have defined a named constant SCENE-HT , to be used as the height of sunset scenes in your program....
-
The employees of Custom Clothes work Monday through Friday. Every other Friday the company issues payroll checks totaling $32,000 (or $3,200 per weekday). The current pay period ends on Friday,...
-
Divide and completely simplify the following: x+5214 x-4 x-49 x-2
-
Define any two of the following terms: Autocratic Leader. Situational leadership model. Leadership style.
-
Considering retail price is 100% consisting both cost and initial markup, a salesperson presented The Wonderland Bookstore, a store that carries Lewis Carroll memorabilia, an opportunity to stock...
-
Rodent Corporation produces two types of computer mice, wired and wireless. The wired mice are designed as low-cost, reliable input devices. The company only recently began producing the...
-
The diagram shows the two forces acting on a small object. Which of the following is the resultant force on the object? A. 8 N downwards B. 8 N upwards C. 2 N downwards D. 2 N upwards 3 N 5 N
-
Draw the structure of the protected amino acid that must be anchored to the solid support in order to use a Merrifield synthesis to prepare leucine enkephalin. (N terminus) Try-Gly-Gly-Phe-Leu (C...
-
Draw all four possible dipeptides that are obtained when a mixture of l-phenylalanine and l alanine is treated with DCC.
-
Show all steps necessary to make the dipeptide Phe-Ala from l-phenylalanine and l-alanine.
-
Why do you think the Unix operating system is a popular attack target by hackers? What should we do to better protect this environment?
-
Your manager wants to implement RBAC functionality. Your shop uses a UNIX system. You do not have a system with an implementation of RBAC available to you. You have the basic capabilities of...
-
You've recently been hired by Me Again, Inc., a successful online retail company that personalizes clothing and accessories with embroidery, screen printing, and laser engraving. The company is ready...

Study smarter with the SolutionInn App


