Use tabulated electrode potentials to calculate G rxn for each reaction at 25 C. 2+ a. 2
Question:
Use tabulated electrode potentials to calculate ΔG°rxn for each reaction at 25 °C.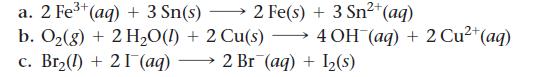
Transcribed Image Text:
2+ a. 2 Fe³+ (aq) + 3 Sn(s) 2 Fe(s) + 3 Sn²+ (aq) b. O₂(g) + 2 H₂O(l) + 2 Cu(s) →→→ 4 OH(aq) + 2 Cu²+ (aq) c. Br₂(1) + 21 (aq) 2 Br (aq) + 1₂(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To calculate Grxn for each reaction at 25 C using tabulated electrode potentialswe can use the follo...View the full answer

Answered By

Vikash Gupta
I am graduated in Physics in 2018, from KIRORIMAL COLLEGE, University of Delhi. Now I am persuing Master's degree in physics. I like to do physics problems. I have experience of 1 year in tutoring. I think Physics is the only subject where you understand things,how they are happening . In physics you learn Maths and apply it. So I would like to join your platform to solve many Physics problems.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use tabulated electrode potentials to calculate G rxn for each reaction at 25 C. a. Pb+ (aq) + Mg(s) b. Br() + 2 CI (aq) c. MnO(s) + 4H+ (aq) + Cu(s) Pb(s) + Mg2+ (aq) 2 Br (aq) + Cl(8) 2+ Mn+ (aq) +...
-
Use tabulated standard electrode potentials to calculate the standard cell potential for the following reaction occurring in an electrochemical cell at 25 C. (The equation is balanced.) Al(s) +...
-
Use the tabulated electrode potentials to calculate G for the reaction Is the reaction spontaneous? I(s) + 2 Br (aq) 21 (aq) + Br(1)
-
Describe at least one method by which the organization can reduce change resistance?
-
Ron LaTulip oversees projects for ACE Construction Company-Recently, the companys controller sent him a performance report regarding the construction of the Campus Highlands Apartment Complex, a...
-
Options can be combined to create more complicated payoff structures. Consider the combination of one put option and one call option with the same expiration date and the same strike price. Draw the...
-
Plaintiffs James and Betty Tonkovich own approximately 850 acres of in Belmont County, Ohio. Plaintiffs belong to a group of landowners known as Belmont Leasing Group, which leases land for oil and...
-
Manufacturing cost data for Orlando Company, which uses a job order cost system, are presented below. Instructions Indicate the missing amount for each letter. Assume that in all cases manufacturing...
-
3. A 200 kg roller coaster starts from rest at the top of the first hill at a height of 20 m above the ground. The second hill is 15 m above the ground. a. A physics student in line for the ride...
-
Calculate the equilibrium constant for each of the reactions in Problem 65. Problem 65 Use tabulated electrode potentials to calculate G rxn for each reaction at 25 C. a. Pb+ (aq) + Mg(s) b. Br() + 2...
-
Which metal is the best reducing agent? a. Mn b. Al c. Ni d. Cr
-
Define each of the following terms in the context of experimental design. a. Response variable b. Factor c. Levels d. Treatments
-
Albany Company has net income before taxes of $90,000, interest expense of $36,000 and an income tax rate of 20%. Based on this information, What is the company's times interest earned ratio ?
-
Write an Oz program which returns the first element of a list and the last element of that list. For example, if the input is [2 ~5 3 ~1 6 ~3 9 4], the output should be 2#4, if the input is[4 ~2],...
-
Why did Pearl River use different entry modes when entering different markets? Pearl River Piano Groups (PRPG) Philosophy is "Music is a journey. And every journey begins with a single step."...
-
A business (with a 31 March balance date) purchases a new vehicle on 20 June. The vehicle has a cost of $30,000 and the depreciation rate (using diminishing value) is 30%. Calculate the depreciation...
-
2. On April 30, Year 2, Wright Enterprise, which sells Business In-Depth Newspapers, receives $1,800 toward newspaper subscription for the next 18 months. Wright Enterprise is a small company and...
-
A copier company has been using the same Copier A for 5 years. This copier can copy approximately 50 sheets a minute. The company has an opportunity to purchase a new Copier B that can process...
-
Find the work done in pumping all the oil (density S = 50 pounds per cubic foot) over the edge of a cylindrical tank that stands on one of its bases. Assume that the radius of the base is 4 feet, the...
-
What is the total number of lines a grating must have in order just to separate the sodium doublet ( 1 = 5895.9 , 2 = 5890.0 ) in the third order?
-
Imagine an opaque screen containing 30 randomly located circular holes. The light source is such that every aperture is coherently illuminated by its own plane wave. Each wave in turn is completely...
-
Perform the necessary mathematical operations needed to arrive at Eq. (10.76). E = (-1)+1 2Kspd (p + r) 2 sin [wt k(p + ro)] (10.76)
-
Michael's father advised him to invest his tuppence instead of spending it, and we calculated that even if he invested it for sixty years he would have only 2.03. However, given a long enough time to...
-
1, x [0,1/4) 1. Suppose f(x) = -3. x 1/4, 3/4). 2. x [3/4,1) (a) Express f in terms of the basis for V2. (b) Decompose f into its component parts in W, Wo, and Vo (i.e., find the Haar decomposition...
-
Use the Gauss-Jordan Algorithm to determine the point of intersection of each system: 3x+5y +7z = 3 (a) x +4y +7z 2x + 2y +3z = 6 Y -2 = 1 y 5 (b) 2x -y -2 = 2 (c) = 2 2 x + y +z = 3 x + y +z = 1

Study smarter with the SolutionInn App


