Use the mass spectrum of mercury to estimate the atomic mass of mercury. Estimate the masses and
Question:
Use the mass spectrum of mercury to estimate the atomic mass of mercury. Estimate the masses and percent intensity values from the graph to three significant figures.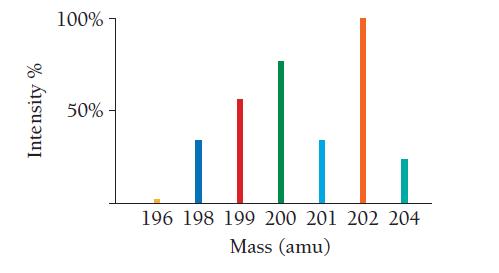
Transcribed Image Text:
Intensity % 100% 50%- 196 198 199 200 201 202 204 Mass (amu)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The mass spectrum of mercury shows two peaksone at 199 amu and one at 201 amuThe peak at 199 am...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the mass spectrum of lead to estimate the atomic mass of lead. Estimate the mass and percent intensity values from the graph to three significant figures. Intensity % 100%- 50%- 204 206 207 208...
-
The masses of the naturally occurring mercury isotopes are 196 Hg, 195.9658 u; 198 Hg, 197.9668 u; 199 Hg, 198.9683 u; 200 Hg, 199.9683 u; 201 Hg, 200.9703 u; 202 Hg, 201.9706 u; and 204 Hg, 203.9735...
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
Suppose that you just purchased 200 shares of Talk&Tell stock for $60 per share. a. If the initial margin requirement is 71.00%, how much money must you borrow? b. Construct the balance sheet...
-
You have $7,000 to deposit. Regency Bank offers 12 percent per year compounded monthly (1 percent per month), while King Bank offers 12 percent but will only compound annually. How much will your...
-
Based on the dimensionless conduction heat rates for cases 12-15 in Table 4.1 b, find shape factors for the following objects having temperature T1, located at the surface of a semi-infinite medium...
-
Using the variance data for Menounos Manufacturing Co., prepare an income statement through gross profit for the year ended December 31, 20Y6. Assume the company sold 3,000 units at $100 per unit.
-
Forten Company, a merchandiser, recently completed its calendar- year 2013 operations. For the year, (1) all sales are credit sales, (2) all credits to Accounts Receivable reflect cash receipts from...
-
1. Assuming firms in a market face a linear downward-sloping demand curve and constant marginal and average costs, carefully explain where equilibrium price would be if it were a competitive market....
-
Pinto company has four employees. In the last week, they have the following work records. The company follows weekly 40 hours or daily 8 hours options to determine Over Time. Overtime is paid @ 1.5...
-
Fill in the blanks to complete the table. Symbol Z A Si 14 S- 2+ Cu+ 15 32 Number Number Number of p of e of n 14 14 15 34 16 Charge 2- 2+
-
Naturally occurring iodine has an atomic mass of 126.9045 amu. A 12.3849 g sample of iodine is accidentally contaminated with an additional 1.00070 g of 129 I, a synthetic radioisotope of iodine used...
-
Explain why all efforts to synthesize a carboxylic acid containing the isotope oxygen-18 at only the carbonyl oxygen fail and yield instead a carboxylic acid in which the labeled oxygen is...
-
A hedge portfolio consists of a dividendpaying stock and a forward contract to eliminate the uncertainty of the selling price of the stock. The forward price should be such that it forces the hedge...
-
Cruiseliners , Inc. has a beta of 1.5 and a cost of equity of 13.2 percent . The risk - free rate of return is 4.2 percent . Cruiseliners is considering a project with a beta of 1.7 and a project...
-
what is the impact on real estate agency if not banking client money into a trust account
-
Review shopifys, expansion activities and business growth of past ten years (approximately) and discuss the following 1. Do you think their selection of international locations/countries was...
-
A father has brought home two equal-sized doughnuts for his children Doug and Isabel. Doug says he loves doughnuts more than Isabel, who agrees, but says she likes them too. The father agrees with...
-
The following information is available for the first month of operations of Beek Inc., a manufacturer of art and craft items: Sales .................. $478,000 Gross profit................ 286,800...
-
Wilsons Auto Repair ended 2011 with Accounts Receivable of $85,000 and a credit balance in Allowance for Uncollectible Accounts balance of $11,000. During 2012, Wilsons Auto Repair had the following...
-
There are five constitutional isomers with molecular formula C 4 H 8 . One of the isomers exhibits a particularly strong signal at M15 in its mass spectrum. Identify this isomer, and explain why the...
-
There are four isomers with molecular formula C 4 H 9 Cl. Only one of these isomers (compound A) has a chirality center. When compound A is treated with sodium ethoxide, three products are formed:...
-
Identify the number of Ï electrons in each of the following compounds. a. b. c. d. e. N'
-
1.Changes in income can impact our consumption choices. As one's income fluctuates, they may also get more joy out of some goods and less from others. Respond to each of the following in 2-3 complete...
-
(b) If the game below is repeated twice, the following strategies form an SPNE when = 1: Strategies Player 1 period 1: Play C period 2: Play B if (C,Y) in stage 1. Play A otherwise. Player 2 period...
-
1. (3 points) Find the outcome of the Borda rule for the following 3-voter profile: >1 >2 >3 b b a a a b d d d Is there a profitable manipulation for the 3rd voter?

Study smarter with the SolutionInn App


