Use the mass spectrum of lead to estimate the atomic mass of lead. Estimate the mass and
Question:
Use the mass spectrum of lead to estimate the atomic mass of lead. Estimate the mass and percent intensity values from the graph to three significant figures.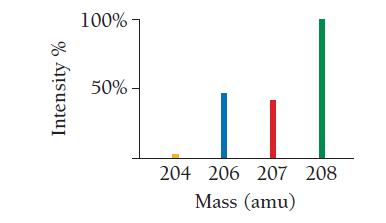
Transcribed Image Text:
Intensity % 100%- 50%- 204 206 207 208 Mass (amu)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
o estimate the atomic mass of lead from the mass spectrum we can take the f...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the mass spectrum of mercury to estimate the atomic mass of mercury. Estimate the masses and percent intensity values from the graph to three significant figures. Intensity % 100% 50%- 196 198...
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
The mass spectrum of an unknown compound has a molecular ion peak with a relative intensity of 43.27% and an M + 1 peak with a relative intensity of 3.81%. How many carbon atoms are in the compound?
-
Show that = E[(m(X) - X'p)] B = argmind (b) = (E[XX']) E[Xm(X)] = [E[XX'])E[XY]. berk Hint: To show E[Xm(X)] = E[XY] use the law of iterated expectations. then
-
You are scheduled to receive $15,000 in two years. When you receive it, you will invest it for six more years at 8 percent per year. How much will you have in eight years?
-
Laser beams are used to thermally process materials in a wide range of applications. Often, the beam is scanned along the surface of the material in a desired pattern. Consider the laser heating...
-
Identify the four components of an ecosystem. After you do this try to visualize the interactions of these four components of an ecosystem as illustrated by Figure 2. 2 in the textbook. Does this...
-
Every home football game for the past eight years at Eastern State University has been sold out. The revenues from ticket sales are significant, but the sale of food, beverages, and souvenirs has...
-
What is the Federal Budget? Explain the process used by the government to create the budget. How does this process differ from the process described by your interviewee who you interviewed in week...
-
In this problem we explore how the potential and kinetic energy of a mass/spring system change in time. In addition to being good practice for energy conservation principles, it will help to set the...
-
Naturally occurring iodine has an atomic mass of 126.9045 amu. A 12.3849 g sample of iodine is accidentally contaminated with an additional 1.00070 g of 129 I, a synthetic radioisotope of iodine used...
-
An a particle, 4 He 2+ , has a mass of 4.00151 amu. Find the value of its charge-to-mass ratio in C/kg.
-
1. Suppose that E and F are mutually exclusive events with Pr(E) = .5 and Pr(F) = .3. Find Pr(E F). 2. Of the 120 students in a class, 30 speak Chinese, 50 speak Spanish, 75 speak French, 12 speak...
-
Ed leaves work for four hours this afternoon and goes to a baseball game. Tickets to the game are $25 and parking at the stadium is $15. Ed earns $15 an hour at his job. What Ed's opportunity cost of...
-
1) How might credit card companies keep their cardholders in debt for a long time? What payment do the credit card companies expect your friend to make so that he never pays down the debt? 2) Can you...
-
21. Determine an equation for the line of intersection of the following planes: 3x + 2y + 5z = 4 and 4x-3y+z = -1. (3 marks)
-
Consider total assets: 750,000 and liabiltiy 750,000 with stock holders equity 27,0000. and Suppose Lehman's commercial real estate is worth 0 with probability of 0.2 and 39.5 billion with...
-
1. Six Sigma is a business process for improving quality. It is favorable for reducing costs and increasing customer satisfaction. DMAIC is a problem-solving process of Six Sigma. Define the 5 steps...
-
Marriott International, Inc., and Wyndham Worldwide Corporation are two major owners and managers of lodging and resort properties in the United States. Abstracted income statement information for...
-
Select the correct answer for each of the following questions. 1. On December 31, 20X3, Saxe Corporation was merged into Poe Corporation. In the business combination, Poe issued 200,000 shares of its...
-
Malonic acid has two acidic protons: The pKa of the first proton (pK 1 ) is measured to be 2.8, while the pK a of the second proton (pK 2 ) is measured to be 5.7. (a) Explain why the first proton is...
-
Identify a systematic (IUPAC) name for each of the following compounds a. b. c. d. (e) CH 3 (CH 2 ) 4 CO 2 H (f) CH 3 (CH 2 ) 3 COCl (g) CH 3 (CH 2 ) 4 CONH 2 O: NH2
-
Identify the common name for each of the following compounds: a. b. c. d.
-
To solve this model economy, we reformulate the competitive equilibrium into the social planner's problem. First of all, in social planner's problem, all markets must clear, and thus N = Nd = N, and...
-
16. Brenda's Balloon Shop is a price taker, and the table below shows its costs of production. Use it to answer the following questions: Output (balloons Total Cost (dollar per hour) per hour) 0 4 1...
-
$400,000 for a new technology.The lender charges them 36% annually with monthly compounding.The agreement calls for no payment until the end of the first month of the 5th year with equal monthly...

Study smarter with the SolutionInn App


