Using the data in Figures 9.15 and 9.16, calculate E for the reaction Na(g) + Cl(g)
Question:
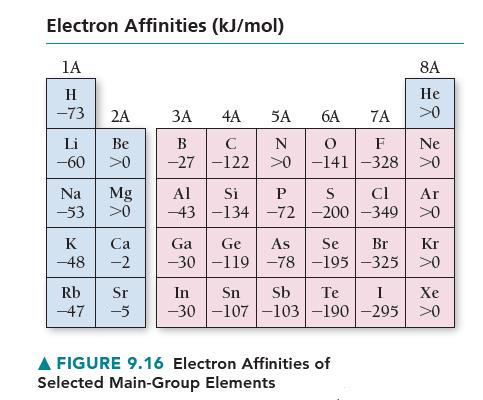
Using the data in Figures 9.15 and 9.16, calculate ΔE for the reaction Na(g)+ Cl(g) → Na+ (g)+ Cl– (g).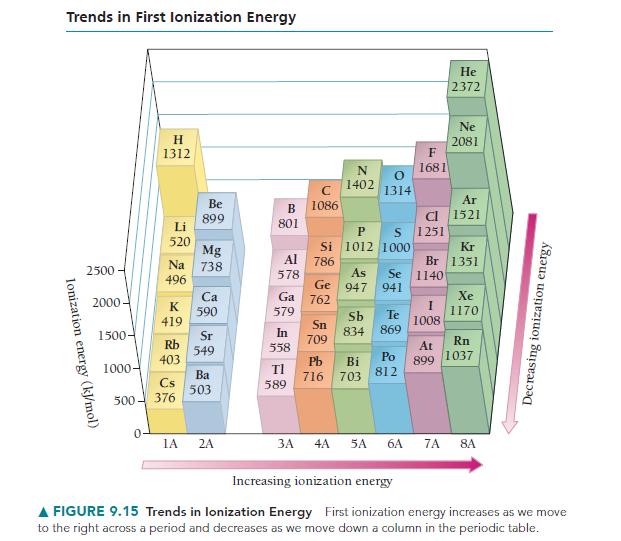
Transcribed Image Text:
Trends in First lonization Energy Ionization energy (kJ/mol) 2500 2000. 1500- 1000- H 1312 0 Li 520 Na 496 Rb 403 Cs 500-376 K 419 Be 899 Mg 738 Ca 590 Sr 549 Ba 503 1A 2A B 801 Al 578 Ga 579 In 558 ΤΙ 589 3A 1086 P Si 1012 786 Ge 762 Sn 709 N 1402 1314 Pb 716 Cl S 1251 1000 As 947 941 Sb Te 834 869 F 1681 Se 1140 Bi Po 703 812 4A 5A 6A I 1008 Kr Br 1351 At 899 He 2372 7A Ne 2081 Ar 1521 Xe 1170 Rn 1037 8A Decreasing ionization energy Increasing ionization energy A FIGURE 9.15 Trends in lonization Energy First ionization energy increases as we move to the right across a period and decreases as we move down a column in the periodic table.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
There are two main terms to consider when calculating E for the reaction Nag Clg Nag Clg th...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using the data in Table 4-3, calculate the G° for ring flip to the other conformation of the molecules depicted in Problem 30. Make sure that the sign (i.e., positive or negative) of your values...
-
Calculate the heat of vaporization, Hovap, of water, using standard enthalpies of formation (Table 6.2). 14270 8 099 0740 9 6416 3818 02015 0 or Hj H Si Si Si Si Si Si A A A A A A So Na Na Na Na Na...
-
A 6 kg block of wood resting on a platform was hit by a 0.08 kg of bullet travelling upward with a speed of 951 m/s. The bullet emerges from the block with a speed of 783 m/s. How much force was...
-
Misty Cumbie worked as a waitress at the Vita Caf in Portland, Oregon. The caf was owned and operated by Woody Woo, Inc. Woody Woo paid its servers an hourly wage that was higher than the states...
-
Willingham Company manufactures raingear. During 2010 Willingham Company decided to issue bonds at 8% interest and then used the cash to purchase a significant amount of treasury stock. The following...
-
A homogeneous wire is bent into the shape shown. Determine by direct integration the x coordinate of its centroid. Express your answer in terms of a. y = kr
-
Consider the net cash flows and salvage values shown below. Assume the alternatives can be indefinitely renewed with the same cash flows and salvage values. Using a MARR of 8%, specify the planning...
-
Brett Dunlop is seeking part-time employment while he attends school. He is considering purchasing technical equipment that will enable him to start a small training services company that will offer...
-
(a) Use the method of direct Integration to find the solution of x2. (b) (i) dy -4x=6. (5%) dx Verify that y = ex, y = e3x and y = Aex + Be -3x (A and B are constants) are - 6y= 0. (10 %) solutions...
-
In reviewing the accounts on September 30, 2020, for the year just ended, Geek Designers discovered the following: a. The September cell phone bill for $465 was unpaid and unrecorded at September 30....
-
Only trace amounts of the synthetic element darmstadtium, atomic number 110, have been obtained. The element is so highly unstable that no observations of its properties have been possible. Based on...
-
A carbon atom can absorb radiation of various wavelengths with resulting changes in its electron configuration. Write orbital diagrams for the electron configuration of carbon that results from...
-
1. If you possess undisclosed inside information, what are your options for trading in the companys stock? 2. How soon after disclosure can an insider trade in the stock? On November 12, 1963, TGS...
-
ALL in R studio Q 1 . 1 . Display the contents of the dataframe. # Do not display the dataframe, just its contents. #Q 2 . 2 . How many cases were sampled in the dataframe? #Q 3 . 3 . How many...
-
Select the scenario depicting a conflict that is solely over values. OOPS Your answer is incorrect. Need more help? Try the Helpful Tutorials. One ethnic group has access to profitable oil fields,...
-
In order to be able to clarify clients' needs in real estate, list three ( 3 ) things you need to do?
-
How is net cash burn calculated? Explain with Example.
-
The full subtractor is a combinational circuit, which is used to perform subtraction of three input bits: the minuend X, subtrahend Y, and borrow in Bin. The full subtractor generates two outputs...
-
The capital investment committee of Overnight Express Inc. is considering two investment projects. The estimated income from operations and net cash flows from each investment are as follows: Each...
-
Why should you not model a decision variable as a random variable with a probability distribution?
-
Consider the couple Ox + e Red with the oxidized and redu ced species at unit activity. What must be the value of E for this half-cell if the reductant Red is to liberate hydrogen at 1 atm from a....
-
By finding appropriate half-cell reactions, calculate the equilibrium constant at 298.15 K for the following reactions: a. 4NiOOH(s) + 2 2 O(l) 4Ni(OH) 2 (s) + O 2 (g) b. 4NO 3 (aq)+ 4H + (aq)...
-
The cell potential E for the cell Pt(s)|H 2 (g, a H2 = 1) H + (aq, a H+ = 1)NaCl(aq, m = 0.300) AgCl(s) Ag(s) is +0.260 V. Determine Cl assuming that = Na+ = Cl .
-
A professor designing a class demonstration connects a parallel-plate capacitor to a battery, so that the potential difference between the plates is 255 V. Assume a plate separation of d = 1.72 cm...
-
A psychrometer has a dry-bulb reading of 90F and a wet-bulb reading of 60F. Find each of the following measurements. (Use the data in this table as necessary.) (a) relative humidity (Enter your...
-
Determine the new value for net income if fixed costs are lowered by 5%, total variable costs rise by 3%, the price is lowered by 5%, and the level of output rises by 10% TFC $3,200,000.00 TVC...

Study smarter with the SolutionInn App


