Which sample of an ideal gas has the greatest pressure? Assume that the mass of each particle
Question:
Which sample of an ideal gas has the greatest pressure? Assume that the mass of each particle is proportional to its size and that all the gas samples are at the same temperature.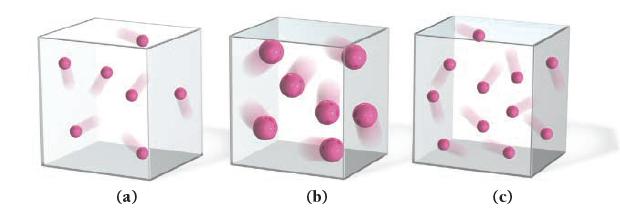
Transcribed Image Text:
(a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
c Since the temperature and the volume are both consta...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(Multiple choice) (1) The temperature change of two blocks of masses MA and MB is the same when they absorb equal amounts of heat. It follows that the specific heats are related by (a) cA =...
-
The town of Beautified is home to many corporations. In order to make sure that it establishes a fair competitive environment for all the City Council wants to pass a law Unfair methods of...
-
Which of the following is not true about an ideal gas? (a) The average kinetic energy of the gas molecules increases as the temperature increases. (b) The volume of an ideal gas increases with...
-
Prepare an Orientation Plan for the select position utilizing the theories discussed in the text. Discuss appropriate and effective orientation plans and your decision-making process for selecting a...
-
Continuous auditing has the potential to reduce labor costs associated with auditing. It also can provide audit assurance closer to the occurrence of a transaction, which improves the reliability of...
-
White Company has two departments, Cutting and Finishing. The company uses a job-order costing system and computes a predetermined overhead rate in each department. The Cutting Department bases its...
-
Quilts R Us (QRU) is considering investing in a new patterning attachment with the cash flow profile shown in the table below. QRU's MARR is 13.5 percent/year. a. What is the internal rate of return...
-
M. Cotteleer Electronics supplies microcomputer circuitry to a company that incorporates microprocessors into refrigerators and other home appliances. One of the components has an annual demand of...
-
What is the difference between a Relation in an RDBMS and a Class in an OODBMS? Are they interchangeable? This question is asking if a relation within a Relational Database Management System is the...
-
A sample of gas has a mass of 0.311 g. Its volume is 0.225 L at a temperature of 55 C and a pressure of 886 mmHg. Find its molar mass. SORT The problem gives you the temperature and pressure of...
-
Explain why people may experience ear pain after a rapid change in altitude.
-
Determine the indicated term for the arithmetic sequence with the first term, a 1 , and common difference, d. Determine a 14 when a 1 = 1/3, d = 2/3.
-
What are three logical components of a business process? Define the functions of each. How do the components interact with one another?
-
Explain how electronic data interchange (EDI) is used to link two companies business processes together.
-
Explain the relationship between the periodic mode and batch processing.
-
What 10 elements are included in the study of AIS?
-
Where are error and exception routines shown on systems flowcharts?
-
On February 17, 2010, a flood destroyed the work in process inventory and half the raw materials inventory of the LRT Company. There was no damage to the finished goods inventory. A physical...
-
Havel says the grocer doesnt believe what is on the sign and indeed, he says the grocers customers will barely notice it. But Havel maintains that the sign serves a specific function. How would you...
-
Apply the moment-distribution method to determine the moment at each joint of the symmetric parabolic haunched frame. Supports A and D are fixed. Use Table 132. The members are each 1 ft thick. E is...
-
Determine the horizontal and vertical displacements at joint ¢of the assembly in Prob. 141. Data From Prob. 141. Determine the stiffness matrix K for the assembly. Take A = 0.5 in 2 and E =...
-
Determine the horizontal displacement of joint and the force in member ¡ Take A = 0.75 in 2 , E = 29(10 3 ) ksi. 2 500 lb 4 ft (3) -3 -31H 4 ft-
-
Describe, in detail, situations in which you should consider referring your client to a different counselor and/or agency. If referral is not an option, what ethical alternatives could you identify...
-
Oakmead is the area head office of a Social Services department in the South of England. Its catchment area deals with around100 000 people. The range of services includes an intake unit, devoted to...
-
On July 1 2022, Bermuda Ltd acquired 60% of share capital of Triangle Ltd for $40,000,000. Equity of Triangle Ltd at the acquisition date was: Share capital General reserve Retained earnings $...

Study smarter with the SolutionInn App


