Assign oxidation numbers to all the atoms in each of the following: a. HNO3 b. CuCl c.
Question:
Assign oxidation numbers to all the atoms in each of the following:
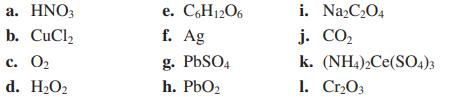
Transcribed Image Text:
a. HNO3 b. CuCl₂ c. 0₂ d. H₂O₂ e. C6H12O6 f. Ag g. PbSO4 h. PbO₂ i. Na₂C₂O4 j. CO₂ k. (NH4)2Ce(SO4)3 1. Cr₂03
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The oxidation numbers of all the atoms in each of the following compounds are a HNO3 H1 N5 O2 b CuCl ...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Assign oxidation numbers to all the atoms in each of the following: a. HNO 3 b. CuCl 2 c. O 2 d. H 2 O 2 e. C 6 H 12 O 6 f. Ag g. PbSO 4 h. PbO 2 i. Na 2 C 2 O 4 j. CO 2 k. (NH 4 ) 2 Ce(SO 4 ) 3 l....
-
Assign oxidation numbers to the atoms in each substance. a. Kr (krypton) b. krypton tetrafluoride (KrF4) c. dioxygen difluoride (O2F2)
-
Assign oxidation numbers to the atoms in each substance. a. Lithium hydride (LiH) b. Potassium peroxide (K2O2) c. Potassium fluoride (KF)
-
I recently heard my neighbor discussing how one of our other neighbors lost his job. My neighbor assumed that the person who lost his job was probably lazy or not smart enough for the job, without...
-
Prepare the journal entries to record these transactions on Kesler Companys books using a periodic inventory system. (a) On March 2, Kesler Company purchased $800,000 of merchandise from Rice...
-
How does absorption costing differ from variable costing? When will absorption costing operating income exceed variable-costing operating income?
-
Titan Offroad Equipment uses the LIFO inventory method and values its inventory using the lower-of-cost-or-market^LCM) rule. Titan Offroad Equipment has the following account balances at December 31,...
-
Miller Enterprises deposits the cash received during each day at the end of the day. Miller deposited $48,287 on October 3 and $50,116 on October 4. Cash register records and other documents...
-
(a) Define capital rationing (4 marks) b) Moyo Kulimba Ltd is considering five projects, V, W, X, Y and Z. Relevant details are as follows: Project Investment required Present value of net NPV Cash...
-
Reba Dixon is a fifth-grade school teacher who earned a salary of $38,000 in 2020. She is 45 years old and has been divorced for four years. She receives $1,200 of alimony payments each month from...
-
In the electrolysis of an aqueous solution of Na 2 SO 4 , what reactions occur at the anode and the cathode (assuming standard conditions)? 2- S0 +2e 250 O + 4H+ + 4e 2HO 2HO + 2e H + 2OH Na + e Na...
-
(a) How is resistance present in all transmission lines? (b) How is inductance present in all transmission lines? (c) How is capacitance present in all transmission lines? (d) The combined effects of...
-
How will the COVID-19 pandemic alter business and society relationships? Identify and discuss some of the major effects and impacts.
-
At Enron executives had incentives to achieve high-revenue growth because their salary increases and cash bonus amounts were linked to reported revenues. In the proxy statement filed in 1997, Enron...
-
How does the role of an export agent differ from the role of an export salesperson?
-
Describe the concepts, principles and constraints underlying financial statements.
-
Soon after the precipitous decline of Arizonas real estate market in 1989, BFA management decided to establish a number of related affiliates. These affiliates were controlled by individuals with...
-
Distinguish between the effect on a CPA firms practice of enforcing the rules of conduct by the AICPA versus a state Board of Accountancy.
-
In the original Western Airlines set-covering model in Figure 14.52, we used the number of hubs as the objective to minimize. Suppose instead that there is a fixed cost of locating a hub in any city,...
-
The activities listed in lines 2125 serve primarily as examples of A) Underappreciated dangers B) Intolerable risks C) Medical priorities D) Policy failures
-
Can a molecule with an inversion center have a dipole moment? Give an example of a molecule with this symmetry element and explain your reasoning.
-
Which of the three normal modes of H 2 O in Figure 27.9 is best described as a bending mode? Does the bond angle remain unchanged in any of the modes? Which requires less energy, bond bending or bond...
-
Why does the list of elements for the D 6h group in Table 27.2 not list the elements C 2 6 , C 3 6 , and C 4 6 ? Selected Point Groups and Their Elements Symmetry Elements TABLE 27.2 Example Molecule...
-
Electronic Arts is contemplating whether its online information webpage should be blue colored or white colored. To answer this question Electronic Arts plans to conduct an A/B test experiment, in...
-
Research your destination about New York City, USA and using secondary data and facts from a variety of sources, and will present this information using the following structure: Title Page Executive...
-
In water cooling tower air enters at a height of 1 m above the ground level and leaves at a height of 7 m. The inlet and outlet velocities are 20 m/s and 30 m/s respectively. Water enters at a height...

Study smarter with the SolutionInn App


