In the electrolysis of an aqueous solution of Na 2 SO 4 , what reactions occur at
Question:
In the electrolysis of an aqueous solution of Na2SO4, what reactions occur at the anode and the cathode (assuming standard conditions)?
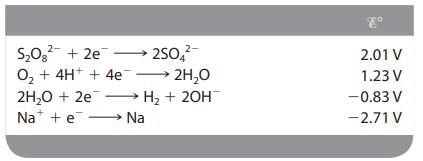
Transcribed Image Text:
2- S₂0² +2e 250 O₂ + 4H+ + 4e →→→ 2H₂O 2H₂O + 2e → H₂ + 2OH Na + e→→→→→→→→→ Na 2.01 V 1.23 V -0.83 V -2.71 V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
In the electrolysis of an aqu...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write equations for the half-reactions that occur at the anode and cathode in the electrolysis of molten KBr. What are the products formed at the anode and cathode in the electrolysis of aqueous KBr?
-
Which product, O 2 or F 2 , is more likely to form at the anode in the electrolysis of an aqueous solution of KF? Explain your reasoning.
-
Predict the products formed in the electrolysis of an aqueous solution of CdSO 4 .
-
Using Figure 7-5 as an example, redraw Figure 7-12 using an enterprise information system that processes a shared database. Explain the advantages of this system over the paper-based system in Figure...
-
On October 5, Lane Company buys merchandise on account from OBrien Company. The selling price of the goods is $5,000, and the cost to OBrien Company is $3,000. On October 8, Lane returns defective...
-
Solve 2u = 2y sin 1/3x for the grid in Fig. 461 and uy (1, 3) = uy (2, 3) = 1/2243, u = 0 on the other three sides of the square.
-
On June 8, 2017, Eugene Weiner made a post on Isaac Aflalos Facebook page. The post read, Yurim and Isaac took advantage of a old 94plus sick man elder abuse [sic]. Alflalo took umbrage to the post...
-
Use Solver to create a Sensitivity Report for question 20 at the end of Chapter 3 and answer the following questions: a. How much would electric trimmers have to cost for the company to consider...
-
Who is Tesla's auditor? Did Tesla's receive a "clean" (unmodified) audit opinion? How many critical audit matters were discussed in Tesla's audit report?
-
Gold is produced electrochemically from an aqueous solution of Au(CN) 2 - containing an excess of CN - . Gold metal and oxygen gas are produced at the electrodes. What amount (moles) of O 2 will be...
-
Assign oxidation numbers to all the atoms in each of the following: a. HNO3 b. CuCl c. 0 d. HO e. C6H12O6 f. Ag g. PbSO4 h. PbO i. NaCO4 j. CO k. (NH4)2Ce(SO4)3 1. Cr03
-
Which of the following is consistent with a steeply upwardly sloping yield curve? a. Monetary policy is expansive and fiscal policy is expansive. b. Monetary policy is expansive while fiscal policy...
-
Find TWO disciplinary outcomes and identify the rules/code of ethics violated and provide the outcome (or checkmark summary): NOT UPCOMING HEARINGS Using...
-
Find a recent short news item that illustrates at least one of the three main types of unemployment (frictional, structural or cyclical). It can be a video link or URL to a current news article...
-
Question 3 (10 marks) A firm is analysing the impact of a price increase on meat. The price was increased from R70 to R78 per kilogramme (kg), resulting in a decline in quantity demanded, from 1 600...
-
You need to do a strategic analysis using the five forces model and the value chain. Which tool should you use first?
-
1. Explain to understand the development of great-power competition in the Arctic, how US foreign policy has developed in the region, and the factors that are impacting this issue. 2. Why Literature...
-
The corporate charter of Luney Corporation allows the issuance of a maximum of 100,000 shares of common stock. During its first two years of operations, Luney sold 70,000 shares to shareholders and...
-
Separate variables and use partial fractions to solve the initial value problems in Problems 18. Use either the exact solution or a computer-generated slope field to sketch the graphs of several...
-
A surface for which the electrostatic potential is negative delineates regions in a molecule that are subject to electrophilic attack. It can help you to rationalize the widely different chemistry of...
-
Hydrocarbons are generally considered to be nonpolar or weakly polar at best, characterized by dipole moments that are typically only a few tenths of a debye. For comparison, dipole moments for...
-
Chemists know that nitric and sulfuric acids are strong acids and that acetic acid is a weak acid. They would also agree that ethanol is at best a very weak acid. Acid strength is given directly by...
-
Ace Construction Company reported the following balance sheet and income statement data for the year ending December 31, 2025. Current assets $ 64,000 Net income $149,000 Current liabilities 24,000...
-
Stuart sues George, Paul, John, and Ringo for negligently injuring him. The jury awards Stuart $1,000,000. The jury also said that Stuart was 25% negligent, George was 10% negligent, Paul was 12%...
-
Pisces Ecclesiastical Ministries (the "Church") is a 45-member congregation operating out of a remote location in California'sSierra Nevada mountainrange. The Church considers fish sacred, and...

Study smarter with the SolutionInn App


