Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution. a.
Question:
Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution.
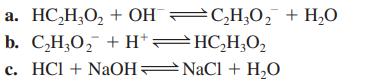
Transcribed Image Text:
a.
HC₂H₂O₂ + OHC₂H₂O₂ + H₂O
b. C,H,O, +H*
a.
HC₂H₂O₂ + OHC₂H₂O₂ + H₂O
b. C,H,O, +H*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Image a chemical equation for a chemical reaction HC2H2O2 OH C2H2O2 H2O The equil...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Calculate the value for the equilibrium constant for each of the following aqueous reactions. a. NH 3 + H 3 O + NH 4 + H 2 O b. NO 2 - + H 3 O + HNO 2 + H2O c. NH 4 + CH 3 CO 2 - NH3 + CH 3 CO 2 H...
-
a. Calculate the value of Ka for the following acids: i. 0.0200 mol dm 3 2-aminobenzoic acid, which has a pH of 4.30 ii. 0.0500 mol dm 3 propanoic acid, which has a pH of 3.10 iii. 0.100 mol dm 3...
-
A firm is engaged in the production of two types of products. The first product (A) earns a profit of $4 per unit and the second product (B) earns $7 per unit. The sales force requires that at least...
-
A trial balance was extracted from the books of V Baker, and it was found that the debit side exceeded the credit side by 40. This amount was entered in the suspense account. The following errors...
-
Richard Vanderbrooks home in New Orleans, Louisiana, was insured through Unitrin Preferred Insurance Co. His policy excluded coverage for, among other things, flood, surface water, waves, tidal...
-
The circuit shown in Fig. contains two batteries, each with an emf and an internal resistance, and two resistors. Find (a) The current in the circuit (magnitude and direction); (b) The terminal...
-
In 1970, Rose Mary Knick purchased 90 acres of land in Scott Township, Lackawanna County, Pennsylvania. In 2008, another resident of Scott Township discovered documents that suggested that one of...
-
Following is a condensed trial balance of the accounts of Leveille Hospital as of December 31, 2013 (amounts in thousands). Additional information (amounts in thousands): 1. The amount of long- term...
-
Determine if the following equations can be solved by separation of variables, and if they are, it solves the problems. 1. 5x7dy+9xy5 = 0 dx 2. -xe +y=0 3. 5xy'-11y = 0 Graph both equations, the...
-
Crude oil has a viscosity of about 0.8 Pas at normal temperature. A 50-km pipeline is to be constructed from an oil field to a tanker terminal. The pipeline is to deliver oil at the terminal at a...
-
Consider the following four titrations (iiv): a. Rank the four titrations in order of increasing pH at the halfway point to equivalence (lowest to highest pH). b. Rank the four titrations in order of...
-
Consider a solution that contains both C 5 H 5 N and C 5 H 5 NHNO 3 . Calculate the ratio [C 5 H 5 N]/[C 5 H 5 NH + ] if the solution has the following pH values: a. pH = 4.50 b. pH = 5.00 c. pH =...
-
Use the nums array to answer. The array was declared using the int nums[4] = {10, 5, 7, 2}; statement. The x and total variables are int variables and are initialized to 0. The avg variable is a...
-
Explain what value there may be in fictionalizing the Netflix show Inventing Anna and in turn how it might explain or illustrate the communicative value and purpose of art. I nclude a discussion of...
-
H. Cochran, Inc., is considering a new three-year expansion project that requires an initial fixed asset investment of $2,350,000. The fixed asset will be depreciated straight- line to zero over its...
-
Two golf balls are hit in the air at 66 feet per second (45 mi/hr), making angles of 37 degrees and 53 degrees with the horizontal. If the ground is level, estimate the horizontal distance traveled...
-
In a 1000 - 1250 word (4-5 page) paper discuss each of the seven family life cycles from the textbook. How important is each stage? What have we learned in this course that can help with...
-
1. Give me a specific example of a time when you usedgood judgment and logic in solving a problem? 2. Tell me a time when you had to carefully analyze a situation in order to be effective in guiding...
-
Wendy's International, Inc., and McDonald's Corporation, two leading fast-food chains, are classified in SIC code 5812-Eating Places. Recent results for each company, along with industry averages,...
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
The acetoacetic ester synthesis cannot be used to make 3,3-dimethyl-2-hexanone. Explain why not.
-
The product of a Dieckmann cyclization can undergo alkylation, hydrolysis, and decarboxylation. This sequence represents an efficient method for preparing 2-substituted cyclopentanones and...
-
Identify the major product formed when each of the following compounds is treated with Et2CuLi followed by mild acid. (a) (b) (c) CN
-
You are tossing pizza dough in the air. You toss the dough straight up in the air with an initial velocity of 5 m/s. a)How high will your pizza dough go? How long will it take to reach its maximal...
-
In the United States, the average age of menarche (firstmenstrual period) is 12.75 years. Now, let's suppose that residentswho live near an abandoned chemical plant are concerned about...
-
In an equal footing state, property owner O owes the following: $160,000 - Mortgage Lender, recorded January 1, 2020 $40,000 - Foundation company, began construction December 30, 2019 $50,000 -...

Study smarter with the SolutionInn App


