Consider a solution of an unknown salt having the general formula BHCl, where B is one of
Question:
Consider a solution of an unknown salt having the general formula BHCl, where B is one of the weak bases in Table 13.3. A 0.10-M solution of the unknown salt has a pH of 5.82. What is the actual formula of the salt?
Table 13.3.
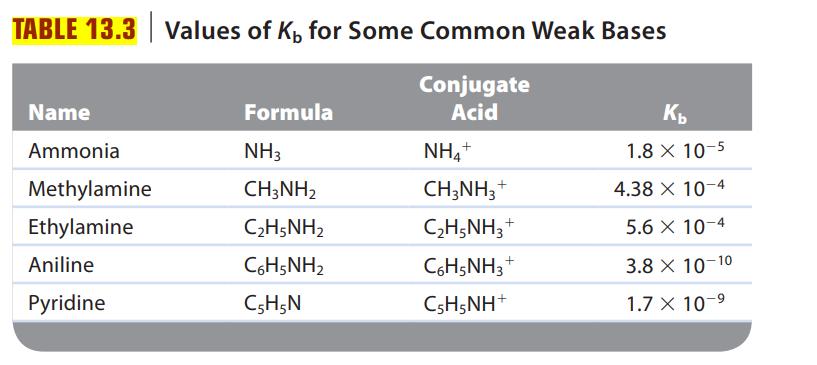
Transcribed Image Text:
TABLE 13.3 Values of K, for Some Common Weak Bases Conjugate Acid Name Ammonia Methylamine Ethylamine Aniline Pyridine Formula NH3 CH3NH₂ C2H5NH2 CoH5NH2 C5H5N NH4+ CH3NH3 + C₂H5NH3 C6H5NH3 CsH5NH* + + Kb 1.8 X 10-5 10-4 10-4 10-10 10-⁹ 4.38 x 5.6 x 3.8 X 1.7 X
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To determine the actual formula of the unknown salt we need to determine the identity of the weak ba...View the full answer

Answered By

Antony Mutonga
I am a professional educator and writer with exceptional skills in assisting bloggers and other specializations that necessitate a fantastic writer. One of the most significant parts of being the best is that I have provided excellent service to a large number of clients. With my exceptional abilities, I have amassed a large number of references, allowing me to continue working as a respected and admired writer. As a skilled content writer, I am also a reputable IT writer with the necessary talents to turn papers into exceptional results.
4.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
An aqueous solution of an unknown salt of gold is electrolyzed by a current of 2.75 amps for 3.50 hours. The electroplating is carried out with an efficiency of 90.0%, resulting in a deposit of...
-
An aqueous solution of an unknown salt of vanadium is electrolyzed by a current of 2.50 amps for 1.90 hours. The electroplating is carried out with an efficiency of 95.0%, resulting in a deposit of...
-
Separate samples of a solution of an unknown salt are treated with dilute solutions of HBr, H2SO4, and NaOH. A precipitate forms in all three cases. Which of the following cations could the solution...
-
If net assets of a business totalled 200,000 and its total assets on that date amounted to 325,000, its liabilities would amount to: (a) 125,000 (b) 200,000 (c) 525,000 (d) Not sufficient information...
-
Vickie Lynn Smith, an actress and model also known as Anna Nicole Smith, met J. Howard Marshall II in 1991. During their courtship, J. Howard lavished gifts and large sums of money on Anna Nicole,...
-
For the circuit shown in Fig. a 20.0-Ω resistor is embedded in a large block of ice at 0.00oC, and the battery has negligible internal resistance. At what rate (in g/s) is this circuit melting...
-
The codon change (Gly-12 to Val-12) in human H-ras that converts it to oncogenic H-ras has been associated with many types of cancers. For this reason, researchers would like to develop drugs to...
-
Clark Kent, Inc., buys crypton for $0.80 a gallon. At the end of processing in Dept. 1, crypton splits off into products A, B, and C. Product A is sold at the split-off point with no further...
-
1. Mr. i holds the following preferences over outcomes w, x, y, and z: xPw, xPy, zPx, yPz, wPy, and wPz. (a) Identify all five possible subsets of these outcomes that contain three or more outcomes....
-
1. Why did VW develop the software in the defeat devices? 2. Has Mllers commitment to maximum transparency helped or hindered the situation? Explain your answer 3. Would you buy a VW car based on the...
-
Calculate the pH of each of the following solutions. a. 0.12 M KNO 2 b. 0.45 M NaOCl c. 0.40 M NH 4 ClO 4
-
Are solutions of the following salts acidic, basic, or neutral? For those that are not neutral, write balanced equations for the reactions causing the solution to be acidic or basic. The relevant Ka...
-
The graphs of y = x 2 (x + 1) and y = 1/x (x > 0) intersect at one point x = r. Use Newtons method to estimate the value of r to four decimal places. 3 2 1 0 y = x2(x + 1) 1 2 +x
-
A company is evaluating a project that requires an initial investment of $80,000 pesos. In the first year, it will generate profits of $30,000, and these earnings will increase by 10% each year. The...
-
A heat pump absorbs 10 kW of heat from outside environment at 250 K while absorbing 15 kW of work. It delivers the heat to a room that must be kept warm at 300 K. The Coefficient of performance (COP)...
-
Check your eLMS for all the topics discussed from prelim to final period. Create a proposal on how you would like to showcase any art as your best practice. The art you choose should be related to...
-
Christian Pawl holds the opinion that the Black Diamond Skylight three-person three-season tent is the most amazing tent on the market. Pawl is so convinced of the tent's versatility that he would...
-
You are preparing for your role as an international marketing manager and want to get a deeper understanding of high context cultures. Explain what a high-context culture is, what are its...
-
In a press release, Logitech, a manufacturer of computer peripherals, announced that sales revenue for the most recent fiscal year had increased 21% over the previous year. If sales revenue for the...
-
How will relating product contribution margin s to the amount of the constrained resource they consume help a company maximize its profits?
-
Propose an efficient synthesis for the following transformation.
-
For a pair of keto-enol tautomers, explain how IR spectroscopy might be used to identify whether the equilibrium favors the ketone or the enol.
-
Acrolein is an α,β-unsaturated aldehyde that is used in the production of a variety of polymers. Acrolein can be prepared by treating glycerol with an acid catalyst. Propose...
-
One of the core classes at Hogwarts is Potions. Three potions taught in this class are the antidote to common poisons, the forgetfulness potion and the Swelling Solution. In order to brew one...
-
When performing trig calculations with the TI graphing calculator make sure you are in the correct mode (degrees or radians). In addition, our calculators don't provide exact answers. Find the six...
-
As part of their final project in AP stats, Christian and Rachel randomly select 18 rolls of toilet paper to measure how well this brand could absorb water. To do this, they poured a 1/4 cup of water...

Study smarter with the SolutionInn App


