Consider the following electrostatic potential diagrams for some covalent compounds. Which of the represented compounds would not
Question:
Consider the following electrostatic potential diagrams for some covalent compounds. Which of the represented compounds would not be soluble in water?
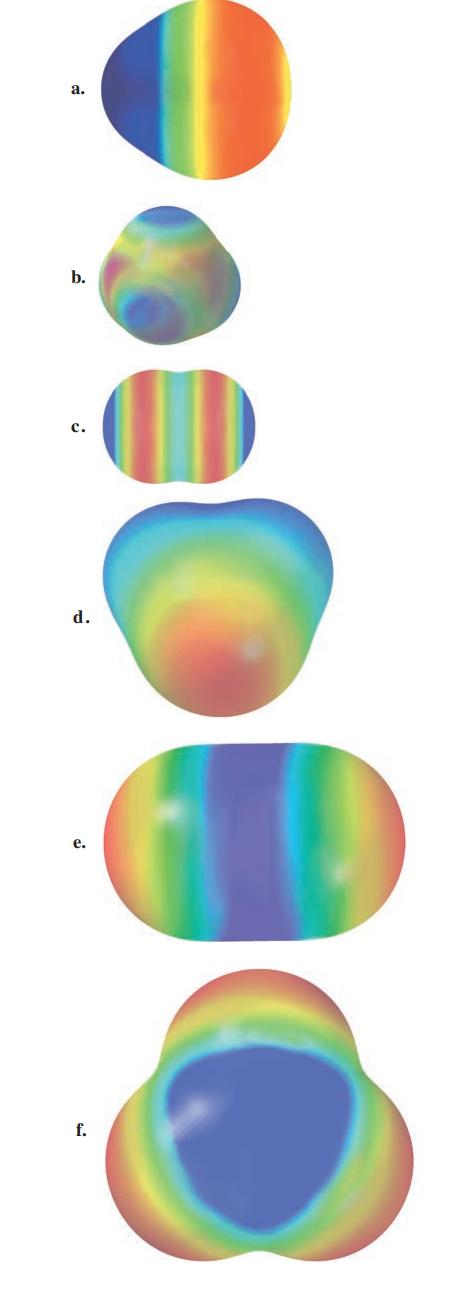
Transcribed Image Text:
a. b. C. d. e. f.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The compounds that are not soluble in water are a b c d e f and g Water is a polar molecule and it i...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Consider the following electrostatic potential diagrams: Rank the compounds from lowest to highest boiling point, and explain your answer. Ethanol Propane Acetone
-
The following electrostatic potential diagrams represent H2, HCl, or NaCl. Label each, and explain your choices. (a) (b)
-
The following electrostatic potential diagrams represent CH4, NH3, or H2O. Label each, and explain your choices. a. C. b.
-
You start driving east for 14 miles, turn left, and drive north for another 10 miles. At the end of driving, what is your straight line distance from your starting point? Round to the nearest tenth...
-
Kelly Malone plans to have $50 withheld from her monthly paycheck and deposited in a savings account that earns 12% annually, compounded monthly. If Malone continues with her plan for two and...
-
Airlines lose thousands of checked bags every day, and America South Airlines is no exception to the industry rule. Over the past 6 weeks, the number of bags misplaced on America South flights has...
-
What do you think led mankind to embark upon the study of Artificial Intelligence? Which fields of study particularly fed into it? What human desires did the study of Artificial Intelligence seek to...
-
The following information has been obtained for the Gocker Corporation. 1. Prior to 2012, taxable income and pretax financial income were identical. 2. Pretax financial income is $1,700,000 in 2012...
-
8. Suppose P is a cubic polyhedron that contains only 4-sided, 6-sided, and 8-sided faces. [10 points] (a) Determine a formula for the number of 4-sided faces in terms of the number of 8-sided faces....
-
Bennett et al. (2003) measured the refractive index (RI) of a pane of glass at 49 di erent locations. She took a sample of 10 fragments at each location and determined the RI for each. The data from...
-
Consider the steps involved in balancing oxidation reduction reactions by using oxidation states. The key to the oxidation states method is to equalize the electrons lost by the species oxidized with...
-
What is an acidbase reaction? Strong bases are soluble ionic compounds that contain the hydroxide ion. List the strong bases. When a strong base reacts with an acid, what is always produced? Explain...
-
In what ways can an operator give the impression of high effort and yet produce at a mediocre or poor level of performance?
-
The internship I applied for is a Sports Marketing and Communications internship with the company Athlitics: What is Athlinics? Our newly imagined sports clinic focuses on inspiring the next...
-
What price do farmers get for their watermelon crops? In the third week of July, a random sample of43farming regions gave a sample mean of= $6.88per 100 pounds of watermelon. Assume thatis known to...
-
Bupa, Australias largest, privately managed health insurance group, merged with the NBF group, Australias second-largest health insurer. A primary objective of the newly established company was to...
-
you are required to answer the following questions. Some questions require a description of the process. Other questions require that you show calculations and answers. What is an outlier and how you...
-
.The vertical distance h travelled by a freely falling body can be computed from the formula h = vot + gt2 where vo is the initial velocity, g is the gravitational acceleration, and t is the time of...
-
Use the chi-square goodness-of-fit to test to determine if the following observed data are normally distributed. Let = .05.What are your estimated mean and standard deviation? Category .... Observed...
-
A 20-cm-square vertical plate is heated to a temperature of 30oC and submerged in glycerin at 10oC. Calculate the heat lost from both sides of the plate.
-
Consider element 113, Nh. What is the expected electron configuration for Nh? What oxidation states would be exhibited by Nh in its compounds?
-
Many lithium salts are hygroscopic (absorb water), but the corresponding salts of the other alkali metals are not. Why are lithium salts different from the others?
-
There are three known xenon fluoride covalent compounds: XeF 2 , XeF 4 , and XeF 6 . In general, the xenon fluoride compounds must be stored in an inert atmosphere, free of oxygen and water. Why is...
-
How do contextual factors, such as organizational culture, task interdependence, and environmental uncertainty, shape team dynamics and performance outcomes, and what organizational interventions are...
-
What are the psychological mechanisms underlying team motivation, including intrinsic motivation, goal setting, and social facilitation, and how can teams cultivate a motivational climate that...
-
Schwenn Products, a bicycle manufacturer, uses normal volume as the basis for setting prices. That is, it sets prices on the basis of long-term volume predictions and then adjusts these prices only...

Study smarter with the SolutionInn App


