Given the following electrostatic potential diagrams, comment on the expected solubility of CH 4 in water and
Question:
Given the following electrostatic potential diagrams, comment on the expected solubility of CH4 in water and NH3 in water.
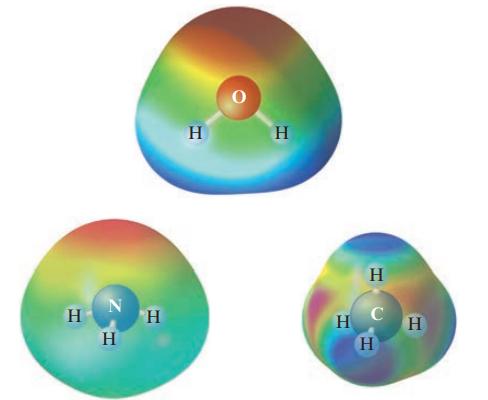
Transcribed Image Text:
н N H Н H Н Н Н с Н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The electrostatic potential maps of CH4 and NH3 show that CH4 is a nonpolar molecule while NH3 is a ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
The following electrostatic potential diagrams represent H2, HCl, or NaCl. Label each, and explain your choices. (a) (b)
-
The following electrostatic potential diagrams represent CH4, NH3, or H2O. Label each, and explain your choices. a. C. b.
-
Consider the following electrostatic potential diagrams for some covalent compounds. Which of the represented compounds would not be soluble in water? a. b. C. d. e. f.
-
The people on Coral Island buy only juice and cloth. The CPI market basket contains the quantities bought in 2016. The average household spent $60 on juice and $30 on cloth in 2016 when the price of...
-
The comparative balance sheet of Flack Inc. for December 31, 2013 and 2012, is shown as follows: The following additional information was taken from the records: a. The investments were sold for...
-
While underdiversification implies lower valuation, entrepreneurs often see ventures as being more valuable than do investors. Why might the entrepreneur value an opportunity more highly?
-
For an ideal gas the change in Gibbs' free energy at constant temperature is given by (a) \(R d \ln T\) (b) \(R T d \ln V\) (c) \(R T d \ln P\) (d) None of these.
-
The stockholders equity T accounts of I-Cards Inc. for the current fiscal year ended December 31, 2014, are as follows. Prepare a statement of stockholders equity for the fiscal year ended December...
-
b. Simulate the graph in given in Figure. 1 to find the shortest path from the graph given below using Dijkstra algorithm, where 'A' is the starting node and 'G' is the destination Value of 'y' is...
-
In terms of Raoults law, distinguish between an ideal liquidliquid solution and a nonideal liquidliquid solution. If a solution is ideal, what is true about H soln , T for the solution formation, and...
-
Some ionic compounds contain a mixture of different charged cations. For example, wstite is an oxide that contains both Fe 2+ and Fe 3+ cations and has a formula of Fe 0.950 O 1.00 . Calculate the...
-
Why do you think there are many different types of charts and graphs?
-
Peter and Doug are neighbors who hate one another. One day, Doug is nailing some boards together on the common sidewalk thathe shares with Peter. In a classic slapstick comedy move, Doug picks up a...
-
b. A bank offers a corporate client a choice between borrowing cash at 9% per annum and borrowing Bitcoin at -5% per annum. The risk-free interest rate is 3% per annum, and storage costs are 6% + (1%...
-
What policies are currently in place to address wealth and housing inequality in Canada? In your opinion, are these policies enough?
-
What was CMS's conclusion regarding the practice restriction in AO- 2011-01? What factors did CMS consider in reaching this conclusion?
-
What role do social movements and collective action play in driving processes of social change, and how do theories of collective behavior, resource mobilization, and political opportunity structures...
-
Workers in several industries were surveyed to determine the proportion of workers who feel their industry is understaffed. In the government sector, 37% of the respondents said they were...
-
What is the expected payoff of an investment that yields $5,000 with a probability of 0.15 and $500 with a probability of 0.85? Select one: O a. $325 O b. $5,500 O c. $2,750 O d. $1,175
-
Propose a mechanism for the following transformation: . 1) Excess MeMgBr 2) H20
-
An ideal gas undergoes an expansion from the initial state described by P i , V i , T to a final state described by P f , V f , T in a. A process at the constant external pressure P f . b. In a...
-
Show the reagents you would use to achieve the following transformation:
-
How do the factors influencing programmed and nonprogrammed decision-making processes differ, and how do these differences impact the effectiveness of decision outcomes?" The article I came across...
-
How do database vendors handle cross-platform compatibility and standardization of triggers, and what are the challenges in developing portable trigger-based logic for different database systems ?
-
Assuming a linear cost relationship, if total costs are $500,000 and variable costs are $30 per unit, what is the amount of fixed costs if production is 15,000 units?

Study smarter with the SolutionInn App


