Order the following solids (ad) from least soluble to most soluble. Ignore any potential reactions of the
Question:
Order the following solids (a–d) from least soluble to most soluble. Ignore any potential reactions of the ions with water.
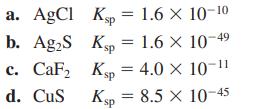
Transcribed Image Text:
a. AgCl Kp = 1.6 × 10-10 Ksp = 1.6 × 10- b. Ag₂S c. CaF₂ d. CuS Ksp = 4.0 × 10-¹¹ Ksp = 8.5 X 10-45
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Note When a salt has a larger Ksp value its solubility will be hi...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Assuming that the solubility of Ca 3 (PO 4 ) 2 (s) is 1.6 10 -7 mol/L at 25 C, calculate the K sp for this salt. Ignore any potential reactions of the ions with water.
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
Part 1 a. Ammonia, NH 3 , is a weak electrolyte. It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion. Write the balanced chemical reaction for this...
-
Perform the indicated operations and simplify the result. Leave your answer in factored form. X x - 3 x + 1 x2 + 5x 24
-
What type of bailment relationship was created when Denai agreed to store Finneys boat? What degree of care was Denai required to exercise in storing the boat? Vanessa Denai purchased forty acres of...
-
Fremont Enterprises has an expected return of 15% and Laurelhurst News has an expected return of 20%. If you put 70% of your portfolio in Laurelhurst and 30% in Fremont, what is the expected return...
-
Match the term with its definition by placing the corresponding letter in the space provided: C Example: Amounts owed to a business by another business or individual 1. A contra-account, related to...
-
The weighted average cost of capital for a firm (assuming all three Modigliani and Miller assumptions apply) is 15 percent. What is the current cost of equity capital for the firm if its cost of debt...
-
Consider a client that has been making a material called UHWDPE (Ultra-high-molecular-weight polyethylene) for hip replacements. They already have an exclusive license to a university patent on the...
-
Fawcett Institute provides one-on-one training to individuals who pay tuition directly to the business and also offers extension training to groups in off-site locations. Fawcett prepares adjusting...
-
A 50.0-mL sample of 0.00200 M AgNO 3 is added to 50.0 mL of 0.0100 M NaIO 3 . What is the equilibrium concentration of Ag + in solution? (K sp for AgIO 3 is 3.0 10 -8 .)
-
Calculate the solubility of Co(OH) 2 (s) (K sp = 2.5 10 -16 ) in a buffered solution with a pH of 11.00.
-
If the S&P/TSX Composite Index declined from 14,614 to 14,238 over a 50-day period, what were the simple and effective annualized rates of decline in the index during the period?
-
What are the classes and patterns involved in defining the stable pattern for temptation?
-
What is the trade-off of using this stable pattern versus the traditional approach?
-
What do you think are the implementation issues for the AnyLevel BO, when used in the temptation stable analysis pattern?
-
What are the possible design issues for the temptation EBT, when linked to the design phase?
-
List a couple of advantages of using the stable analysis pattern for temptation.
-
Economists assume that our wants are unlimited. How does that assumption relate to the basic questions relating to economics?
-
Wimot Trucking Corporation uses the units-of-production depreciation method because units-of-production best measures wear and tear on the trucks. Consider these facts about one Mack truck in the...
-
Identify the reagents that you would use to accomplish each of the following transformations: (a) (b) H.
-
Predict the major product for each of the following transformations: (a)
-
Identify the reagents that you would use to accomplish each of the following transformations (you will also need to use reactions from previous chapters). (a) (b) (c) Br Br OH
-
Translate the following C code function into ARM assembly language. int32_t g32(int32_t x) { int32_t f32(int32_t); return xf32(x+9); }
-
1. Consider the DTD below for an airline data. Give an (non-trivial) example XML document that conforms to the above DTD.
-
Green eyes (E) is dominant in a species of iguanas. Blue eyes is recessive. By Mendelian convention, the recessive allele for eye color is labeled _-. genotype? If an iguana is homozygous recessive...

Study smarter with the SolutionInn App


