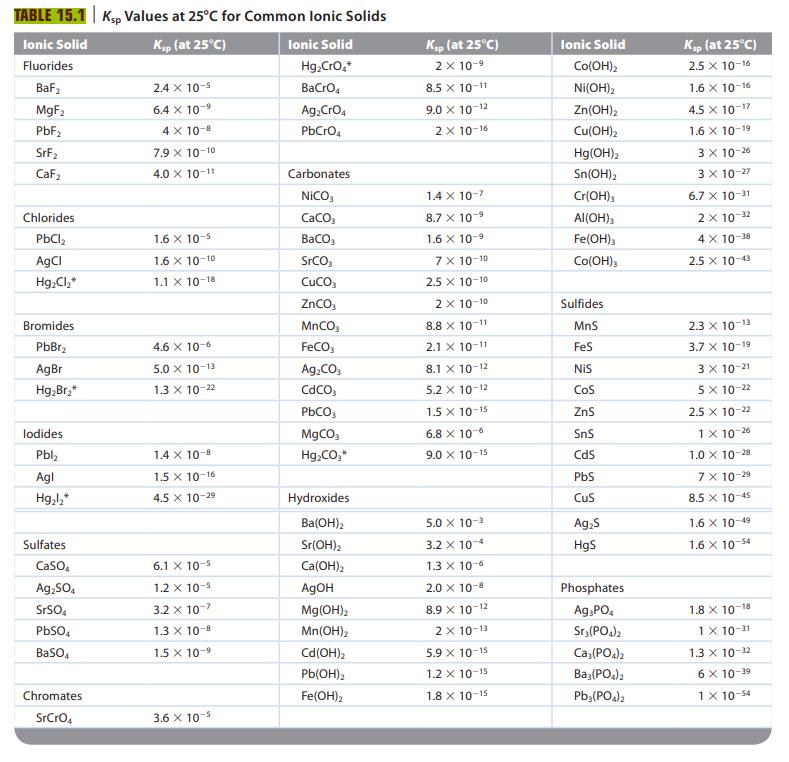
To what reaction does the solubility product constant, Ksp, refer? Table 15.1 lists K sp values for
Question:
To what reaction does the solubility product constant, Ksp, refer? Table 15.1 lists Ksp values for several ionic solids. For any of these ionic compounds, you should be able to calculate the solubility. What is the solubility of a salt, and what procedures do you follow to calculate the solubility of a salt? How would you calculate the Ksp value for a salt given the solubility?
Transcribed Image Text:
TABLE 15.1 Ksp Values at 25°C for Common Ionic Solids Ionic Solid Ksp (at 25°C) Fluorides BaF₂ MgF₂ PbF₂ SrF₂ CaF₂ Chlorides PbCl₂ AgCl Hg₂Cl₂* Bromides PbBr₂ AgBr Hg₂Br₂* lodides Pbl₂ Agl Hg₂l₂* Sulfates CaSO4 Ag₂SO4 SrSO4 PbSO4 BaSO4 Chromates SrCrO4 2.4 x 10-5 6.4 x 10-9 4 X 10-8 7.9 X 10-10 4.0 X 10-11 1.6 X 10-5 1.6 X 10-10 1.1 X 10-18 4.6 x 10-6 5.0 X 10-13 1.3 x 10-22 1.4 x 10-8 1.5 X 10-16 4.5 X 10-2⁹ 6.1 x 10-5 1.2 x 10-5 3.2 x 10-7 1.3 X 10-8 1.5 X 10-9 3.6 x 10-5 Ionic Solid Hg₂CrO,* BaCrO4 Ag₂ CrO4 PbCrO4 Carbonates NICO, CaCO3 BaCO3 SrCO CUCO3 ZnCO₂ MnCO₂ FeCO3 Ag₂CO3 CdCO PbCO3 MgCO3 Hg₂CO,* Hydroxides Ba(OH)2 Sr(OH)2 Ca(OH)2 AgOH Mg(OH)2 Mn(OH)₂ Cd(OH)₂ Pb(OH)2 Fe(OH)₂ Ksp (at 25°C) 2 x 10-⁹ 8.5 X 10-11 9.0 X 10-12 2 X 10-16 1.4 x 10-7 8.7 X 10 -9 1.6 × 10-9 7 x 10-10 2.5 X 10-10 2 x 10-10 8.8 x 10-11 2.1 X 10-11 8.1 X 10-12 5.2 x 10-12 1.5 X 10-15 6.8 x 10-6 9.0 X 10-15 5.0 × 10-³ 3.2 x 10-4 1.3 x 10-6 2.0 × 10-8 8.9 X 10-12 2x 10-13 5.9 X 10-15 1.2 X 10-15 1.8 X 10-15 Ionic Solid Co(OH)2 Ni(OH)2 Zn(OH)₂ Cu(OH)₂ Hg(OH)2 Sn(OH)2 Cr(OH)3 Al(OH)3 Fe(OH)3 Co(OH)3 Sulfides MnS FeS NIS COS ZnS SnS CdS PbS CuS Ag₂S HgS Phosphates Ag3PO4 Sr3(PO4)2 Ca3(PO4)2 Ba3(PO4)2 Pb3(PO4)2 Ksp (at 25°C) 2.5 X 10-16 1.6 X 10-16 4.5 X 10-1 1.6 X 10-19 3 x 10-26 3 x 10-27 6.7 X 10-31 2 x 10-32 4 X 10-38 2.5 X 10-43 2.3 X 10-13 3.7 X 10-19 3 x 10-21 5 x 10-22 2.5 x 10-22 1 X 10-26 1.0 X 10-28 7 x 10-29 8.5 x 10-45 1.6 X 10-49 1.6 x 10-54 1.8 X 10-18 1 X 10-31 1.3 x 10-32 6 x 10-39 1 x 10-54
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Lets figure out how soluble BaCrO 4 is Utilizing the supplied K sp value from Table 151 which i...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write a project management plan. we have a template and project description. we need to edit the template(table of contents) with our own ideas. CPSC 8820-01 Project Management Plan Your Unique...
-
Read the article " The Twitter Trap " by Bill Keller carefully. In this article, Keller uses his personal experiences to attract the audience and uses funny examples to help the audience understand...
-
Calculate the solubility product constant for Mg(OH)2 at 25oC. The Gof values (in kJ/mol) are as follows: Mg2+ (aq), 454.8; OH(aq), 157.3; Mg(OH)2(s), 833.7.
-
State whether or not each of the following events would result in a liability being recognised in the accounts at 30 June. 1. Taxes for the year ended 30 June, which are not payable until October. 2....
-
Go to this texts Web site at academic. cengage.com/blaw/clarkson and select Chapter 49. Click on Video Questions and view the video titled Double Indemnity. Then answer the following questions. (a)...
-
A current-carrying gold wire has diameter 0.84 mm. The electric field in the wire is 0.49 V/m. What are? (a) The current carried by the wire; (b) The potential difference between two points in the...
-
Increasingly, we are seeing email used in cases involving defendants located in foreign countries. Plaintiffs filed suit against four Defendants: Qingdao Sunflare New Energy Co., Skone Lighting Co.,...
-
The following events apply to Montgomery Company for 2016, its first year of operation: 1. Received cash of $36,000 from the issue of common stock. 2. Performed $48,000 of services on account. 3....
-
Stage duration (hours) for immatures of 2 species of blow fly when reared at several temperatures; e.g., "time to complete" the stage. The threshold temperature for all species is 10C.--------1. What...
-
1. As much as possible, assess the extent to which PetroChinas accounting principles conform to IASB standards. 2. How reliable is your assessment? 3. What further information would help your...
-
Fig. 15.2 summarizes the classic method for separating a mixture of common cations by selective precipitation. Explain the chemistry involved with each of the four steps in the diagram. Fig. 15.2...
-
Consider the following four titrations (iiv): a. Rank the four titrations in order of increasing pH at the halfway point to equivalence (lowest to highest pH). b. Rank the four titrations in order of...
-
Garcia Company issues 10%, 15-year bonds with a par value of $240,000 and semiannual interest payments. On the issue date, the annual market rate for these bonds is 8%, which implies a selling price...
-
How can acting unprofessionally in a business social situation (for example the annual company picnic or holiday party) hurt your career? Share some examples of inappropriate behavior at business...
-
What would be the appropriate adjusting journal entries: The notes payable balance relates to a bank loan obtained in 2018 that is payable in full on august 31,2023. the loan agreement specifies that...
-
1:Explain your understanding of why it is important to be persuasive in negotiation and how you would go about being persuasive in a negotiation. 2: Research a conflict or dispute that has occurred...
-
What is going on with your site? I have searched for examples for the following papers and could not find any info on them. They are: 1. Create Visual Communication 2. Comm Regulation and Policy 3....
-
One of your staff members has been asked to give two speeches. Details are as follows: first speech - large target audience; objective: to inform them of company progress over the past year; and ...
-
Pippin Piping Co. manufactures decorative fabric trims. The company's financial statements follow. Required a. Prepare a horizontal analysis for 2013. What significant changes do you see? b. Prepare...
-
When the Department of Homeland Security created a color-coded system to prepare government officials and the public against terrorist attacks, what did it do right and what did it do wrong?
-
Identify the reagents you would use to achieve the following transformation:
-
Propose an efficient synthesis for each of the following compounds using the malonic ester synthesis. (a) (b) (c) (d) (e)
-
Starting with diethyl malonate and using any other reagents of your choice, proposean efficient synthesis for each of the following compounds: (a) (b) (c) .
-
3. A standard piece of printer paper has a perimeter of 39 inches. A standard sized piece of paper has dimensions of 8.5 inches by 11 inches, but does this maximize the area of the paper? Your goal...
-
Serena Williams is one the best tennis player in the world. Shehas won 23 grand slam titles. She has one of the fastest serve inTennis. Her serve has been clocked at 177 kilometers per hour. Whatis...
-
The University of Washington bookstore is concerned about losing customers due to bad service. It decides to compute a lifetime value of the "impoverished" customer to communicate the importance of...

Study smarter with the SolutionInn App


