Acrolein, C 3 H 4 O, is the starting material for certain plastics. (a) Which bonds in
Question:
Acrolein, C3H4O, is the starting material for certain plastics.
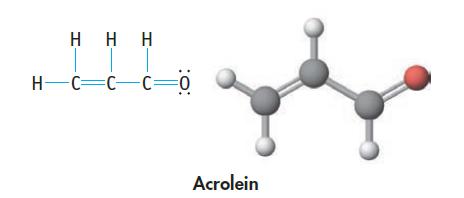
(a) Which bonds in the molecule are polar, and which are nonpolar?
(b) Which is the most polar bond in the molecule? Which is the more negative atom of this bond?
Transcribed Image Text:
Η Η Η H=C=C=C=0 Acrolein
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a The polar bonds in acrolein are the CO bond and the CO bond The nonpolar bond is the CC bond b The ...View the full answer

Answered By

Ajay Negi
Hi, I've completed my degree in engineering (Information Technology) from an NIT. Currently working as a software engineer. Wish to impart quality education to the future generation.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
The starting material for a commercial synthesis of vitamin C is l-sorbose (see the following reaction); it can be synthesized from d-glucose through the following reaction sequence: The second step...
-
Suppose the relationship between wage, years of education (educ), years of experience (exper), and participation in a job training program (train) is modeled as: log ( ) = + + + . Which of the...
-
37) Because of inability to pay its debts, the Lou Gi Manufacturing Company has been forced into bankruptcy as of April 1, 20x7. The balance sheet on the date shows: Assets Cash Accounts Receivable...
-
What did the company do right that has led to the successful deployment of data warehousing and business intelligence within Norfolk Southern? Norfolk Southern Railway Company operates one of the...
-
A farm implements dealer is seeking a fourth warehouse location to complement three existing warehouses. There are three potential locations: Charlotte, NC; Atlanta, GA; and Columbia, SC. Charlotte...
-
A computer virus is a kind of intelligent agent .Discuss this statement. Consider the various agent properties that have been discussed in this chapter. Which of these properties do computer viruses...
-
Due to the poor quality of various semiconductor products used in their manufacturing process, Microlaboratories has decided to develop a QC program. Because the semiconductor parts they get from...
-
Types of recruitment In the past, the recruitment process was a reactive one in which companies did not necessarily conduct an analysis of the type of strategy they would use to recruit candidates....
-
Urea, (NH 2 ) 2 CO, is used in plastics and fertilizers. It is also the primary nitrogen-containing substance excreted by humans. (a) Which bonds in the molecule are polar, and which are nonpolar?...
-
For each pair of bonds, indicate the more polar bond, and use an arrow to show the direction of polarity in each bond. (a) CO and CN (b) PBr and PCl (c) BO and BS (d) BF and BI
-
Johnson Corporation began 2013 with inventory of 10,000 units of its only product. The units cost $8 each. The company uses a periodic inventory system and the LIFO cost method. The following...
-
On January 1, 2024, Majestic Mantles leased a lathe from Equipment Leasing under a finance lease. Lease payments are made annually. Title does not transfer to the lessee and there is no purchase...
-
What is the probability of developing and dying from cancer today?
-
You are a supervisor at your company. You've overheard Employee A making offensive comments to other employees, and Employee B has complained about these comments. You've given Employee A a verbal...
-
Were the public relations tactics of the food industry ethical? Why or why not? I am always amazed when I watch a documentary about something I think I already know, then learn so much more. The role...
-
What characteristic best aligns with the leadership trait of sociability ?
-
How far do professional golfers drive a ball? (For non-golfers, the drive is the shot hit from a tee at the start of a hole and is typically the longest shot.) Heres a histogram of the average...
-
After graduating from college and working a few years at a small technology firm. Preet scored a high-level job in the logistics department at Amex Corporation. Amex sells high-quality electronic...
-
For a two-dimensional harmonic oscillator, V (x, y) = k x x 2 + k y y 2 Write an expression for the energy levels of such an oscillator in terms of k x and k y .
-
A gas-phase 1 H 127 I molecule, with a bond length of 160.92 pm, rotates in three dimensional space. a. Calculate the zero point energy associated with this rotation. b. What is the smallest quantum...
-
In this problem you will derive the commutator [lx, ly] = ihlz. a. The angular momentum vector in three dimensions has the form l = il x + jl y + kl z , where the unit vectors in the x, y, and z...
-
Define workplace bullying. Describe how anger and aggression are utilized by the bully in workplace bullying. Describe an example of workplace bullying that you are personally aware of or have...
-
Where is the intersection between Leadership and ethics? this is a very broad question.Where does the responsibility to behave ethically when working with older people sit? If a nurse in a nursing...
-
Analyse the business 'Nike', identifying the company's core competencies, resource capabilities and outcomes. Demonstrate an in-depth knowledge and practical application of key business theories,...

Study smarter with the SolutionInn App


