An unknown solid acid is either citric acid or tartaric acid. To determine which acid you have,
Question:
An unknown solid acid is either citric acid or tartaric acid. To determine which acid you have, you titrate a sample of the solid with aqueous NaOH and from this determine the molar mass of the unknown acid. The appropriate equations are as follows: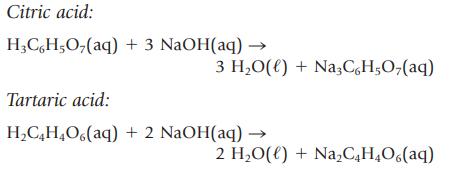
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:





