Balance the following equations: Data given in Example 3.1 (a) Cr(s) + Cl(g) CrCl3(s) (b) SiO
Question:
Balance the following equations:
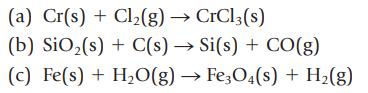
Data given in Example 3.1

Transcribed Image Text:
(a) Cr(s) + Cl₂(g) → CrCl3(s) (b) SiO₂ (s) + C(s) → Si(s) + CO(g) (c) Fe(s) + H₂O(g) → Fe3O4(s) + H₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Crs 3 Cl2g 2 CrCl3s b SiO2s 3 Cs 2 ...View the full answer

Answered By

Jinah Patricia Padilla
Had an experience as an external auditor in Ernst & Young Philippines and currently a Corporate Accountant in a consultancy company providing manpower to a 5-star hotel in Makati, Philippines, Makati Diamond Residences
5.00+
120+ Reviews
150+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The Government of the Republic of Indonesia plans to issue new SBN. SBN are securities issued by the government to finance the state budget and can be an investment instrument for holders (investors)...
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
Majan International School, Muscat conducts online classes and online exams for its students due to Covid 19 pandemic situation. It uses its own online Learning Teaching Management system namely...
-
Explain what the hybrid aggregate plan is and why it is used.
-
Young Professional magazine was developed for a target audience of recent college graduates who are in their first 10 years in a business/professional career. In its two years of publication, the...
-
Water is siphoned from a large tank and discharges into the atmosphere through a 2-in.-diameter tube as shown in Fig. P3.95. The end of the tube is \(3 \mathrm{ft}\) below the tank bottom, and...
-
GAAS, GAGAS, and the Single Audit. A city has approached you concerning the audit of its 2011 financial statements. State law requires the city to have an audit and submit the audited financial...
-
used soon at Insmqclavab inemagenam o dosque osmolya smenil vd ebay abrismeb ener ter ne 2.1 Human Resource (HR) Managers or Training & Development Specialists need to confirm that the content of the...
-
Balance the following equations, and name each reactant and product: Data given in Example 3.1 (a) FeO3(s) + Mg(s) MgO(s) + Fe(s) (b) AlCl3(s) + NaOH(aq) AI(OH)3 (s) + NaCl(aq) (c) NaNO3(s) +...
-
Balance the following equations: (a) Cr(s) + O 2 (g) n Cr 2 O 3 (s) (b) Cu 2 S(s) + O 2 (g) n Cu(s) + SO 2 (g) (c) C 6 H 5 CH 3 ()+ O 2 (g) n H 2 O() + CO 2 (g) Data given in Example 3.1 EXAMPLE 3.1...
-
1. What triggers the conflict in this scene? 2. Does mission commander Jim Lovell successfully manage the group dynamics to return the group to a normal state? This film re-creates the heroic efforts...
-
Three-lens systems. In the figure, stick figure O (the object) stands on the common central axis of three thin, symmetric lenses, which are mounted in the boxed regions. Lens 1 is mounted within the...
-
Describe how a distribution of incentive payments under a Medicaid ACO can potentially implicate the Stark Law?
-
A driver is traveling 68 mi/h on a road with a -3% grade. There is a stalled car on the road 1000 ft ahead of the driver. The driver's vehicle has a braking efficiency of 90% and anti-lock brakes....
-
A marketable security is initially classified as a trading security, an available - for - sale security, or a held - to - maturity debt security. Subsequently, a security can be transferred among...
-
Many students are surprised to learn that contracts made orally are generally enforceable, unless the subject of the agreement falls within the scope of the Statute of Frauds or another statutory...
-
What is the most common constraint faced by merchandisers?
-
Differentiate. y = ln(3x + 1) ln(5x + 1)
-
Use resonance structures to help you identify all sites of low electron density (δ+) in the following compound:
-
Liquid N 2 has a density of 875.4 kg m 3 at its normal boiling point. What volume does a balloon occupy at 298 K and a pressure of 1.00 atm if 3.10 10 3 L of liquid N 2 is injected into it? Assume...
-
Calculate the volume of all gases evolved by the complete oxidation of 0.375 g of the amino acid alanine NH 2 CHCH 3 COOH if the products are liquid water, nitrogen gas, and carbon dioxide gas, the...
-
Simplify. 3x 2 5 3 2x y 3
-
Solve the equation -16x + 4y = 32 for y.
-
Adjusting entries for unearned revenue: decrease liabilities and increase revenues. increase liabilities and increase revenues. increase assets and increase revenues. decrease revenues and...

Study smarter with the SolutionInn App


