Balance the following equations, and name each reactant and product: Data given in Example 3.1 (a) FeO3(s)
Question:
Balance the following equations, and name each reactant and product: 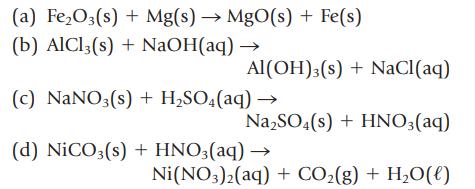
Data given in Example 3.1

Transcribed Image Text:
(a) FeO3(s) + Mg(s) MgO(s) + Fe(s) (b) AlCl3(s) + NaOH(aq) AI(OH)3 (s) + NaCl(aq) (c) NaNO3(s) + HSO4(aq) NaSO4(s) + HNO3(aq) (d) NiCO3(s) + HNO3(aq) Ni(NO3)2(aq) + CO(g) + HO(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Balance and name the reactants and products a Fe 2 O 3 s 3 Mgs 3 MgOs 2 Fes 1 Note the need for at l...View the full answer

Answered By

Hillary Waliaulah
As a tutor, I am that experienced with over 5 years. With this, I am capable of handling a variety of subjects.
5.00+
17+ Reviews
30+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations, and name each reactant and product: Data given in Example 3.1 (a) SF4(g) + HO(l) SO(g) + HF(e) (b) NH3(aq) + O(aq) NO(g) + HO(l) (c) BF3(g) + HO(l) HF(aq) +...
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
The registers values in AX and BX can be exchanged using stack operations that are: Select one: a. PUSH bx PUSH ax POP bx POP AX b. none C. PUSH ax PUSH bx POP ax POP bx d. PUSH ax POP ax PUSH bx POP...
-
Explain the procedure for developing an aggregate plan.
-
The Wall Street Journal reported on several studies that show massage therapy has a variety of health benefits and it is not too expensive. A sample of 10 typical one-hour massage therapy sessions...
-
Water flows in the system shown in Fig. P3.96. Assume frictionless flow. (a) Calculate the rate \(Q\) at which water must be added at the inlet to maintain the 16-ft height. (b) Calculate the height...
-
A 10-year, 12% semiannual coupon bond with a par value of $1,000 may be called in 4 years at a call price of $1,060. The bond sells for $1,100. (Assume that the bond has just been issued.) a. What is...
-
How would you conduct a diagnosis of the situation at HealthCo? Provide examples of the types of data you would collect, how you would collect it, and how you would use it ?
-
Write balanced chemical equations for the following reactions. (a) The reaction of aluminum and iron(III) oxide to form iron and aluminum oxide (known as the thermite reaction). (b) The reaction of...
-
Balance the following equations: Data given in Example 3.1 (a) Cr(s) + Cl(g) CrCl3(s) (b) SiO (s) + C(s) Si(s) + CO(g) (c) Fe(s) + HO(g) Fe3O4(s) + H(g)
-
We consider the field E = Q(2, 3, 5). It can be shown that [E : Q] = 8. In the notation of Theorem 48.3, we have the following conjugation isomorphisms (which are here automorphisms of E): For...
-
Consider the following code segment: = double a 1.0%; = double b 3.0; double output = Math.pow(b, Math.abs (b)) * Math.sq rt(a); System.out.println(output); What is printed when this code segment is...
-
Tropic Treasures Inc reported the following information for their year - ended June 3 0 , 2 0 2 3 . Assume normal balances. Accounts Payable 1 , 8 0 0 Accounts Receivable 3 , 5 0 0 Cash 5 8 , 0 0 0...
-
The partners are unsure as to whether the interest on the loan from Locke should be accounted for in the Income Statement or in the Profit and Loss Appropriation Account. Explain to them how it...
-
Hometowne paid a local radio station $ 1 4 , 2 0 0 for four months of advertising on December 1 , 2 0 2 4 . The advertising will appear evenly over the four - month period. The company debited...
-
Aram sold a capital asset that he owned for more than one year for a $ 2 , 5 6 0 loss, a capital asset that he owned for six months for a $ 4 , 3 2 0 gain, and a capital asset he owned for two months...
-
What is outsourcing?
-
Determine the values of the given trigonometric functions directly on a calculator. The angles are approximate. tan 0.8035
-
Carbon monoxide competes with oxygen for binding sites on the transport protein hemoglobin. CO can be poisonous if inhaled in large quantities. A safe level of CO in air is 50. parts per million...
-
The total pressure of a mixture of oxygen and hydrogen is 1.65 atm. The mixture is ignited and the water is removed. The remaining gas is pure hydrogen and exerts a pressure of 0.190 atm when...
-
Suppose that you measured the product PV of 1 mol of a dilute gas and found that PV = 24.35 L atm at 0.00C and 33.54 L atm at 100.C. Assume that the ideal gas law is valid, with T = t(C) + a, and...
-
Simplify by first factorising the numerator and denominator: x-6x-27 x-9 4x + 12x 4
-
Find all zeros of f(x) = -10/8x+12+12.
-
Find the two possible values of A, A1 and A2 satisfying thefollowing determinantal equation: a- b D = = 0 C d-> for a = 4.1, b = 5.4, c = 4.2 and d = -3.3 and then give, as your answer, the value of...

Study smarter with the SolutionInn App


