Complete and balance the equations below, and classify them as precipitation, acidbase, gasforming, or oxidationreduction reactions. Show
Question:
Complete and balance the equations below, and classify them as precipitation, acid–base, gasforming, or oxidation–reduction reactions. Show states for reactants and products (s, ℓ, g, aq).
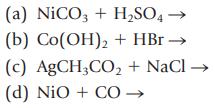
Transcribed Image Text:
(a) NiCO3 + H₂SO4 - (b) Co(OH)2 + HBr → (c) AgCH3CO₂ + NaCl → (d) NIO + CO →
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a NiCO3 s H2SO4 aq NiSO4 aq H2CO3 aq This is an acidbase reaction Carb...View the full answer

Answered By

Lalit Kumar
I love to teach the students.It's just not a profession for me, it is my passion. I had taught many students at the best of my level. I'll give one hundred percent to your problems. Let experience my tutoring skill together.LETS GO.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Complete and balance the equations for the following reactions. a. Li(s) + N2(g) b. Rb(s) + S(s) c. Cs(s) + H2O(l) d. Na(s) + Cl2(g)
-
The following reactions occur in aqueous solution. Complete and balance the molecular equations using phase labels. Then write the net ionic equations. a. CaS + HBr b. MgCO3 + HNO3 c. K2SO3 + H2SO4
-
The following reactions occur in aqueous solution. Complete and balance the molecular equations using phase labels. Then write the net ionic equations. a. BaCO3 + HNO3 b. K2S + HCl c. CaSO3(s) + HI
-
Consider the following actual FY2019 data and a forecast of FY2020 selected balance sheet and income statement numbers. $ millions FY2019 Actual FY2020 Est. $29,009 $32,102 14,592 16,051 8,755 9,923...
-
Figure shows that Japans short-term interest rates have had periods during which they are near or equal to zero. Is the fact that the yen interest rates shown never drop below zero a coincidence, or...
-
Craig Industries was in the business of manufacturing charcoal. Craig, the corporation's president, contracted in the name of the corporation to sell the company's plants to Husky Industries. Craig...
-
Verify that the Tweedie distribution is a member of the linear exponential family of distributions by checking equation (13.9). In particular, provide an expression for \(S(y, \phi)\) (note that...
-
During the most recent year, Butler paid $95,000 in interest to its lenders and $80,000 in dividends to its stockholders. Required 1. In which category of the statement of cash flows (operating,...
-
2) Use the table below for answering the following questions. Questions a through e can be done on paper. You must show your work to support your results. Use Microsoft Project to complete question f...
-
The products formed in several reactions are given below. Identify the reactants (labeled x and y) and write the complete balanced equation for each reaction. (a) x + y HO(l) + CaBr(aq) (b) x + y ...
-
Balance each of the following equations, and classify them as precipitation, acidbase, gas-forming, or oxidationreduction reactions. Show states for reactants and products (s, , g, aq). (a) CuCl + HS...
-
The stockholders equity accounts of Port Corporation on January 1, 2011, were as follows. Preferred Stock (8%, $50 par cumulative, 10,000 shares authorized) . $ 400,000 Common Stock ($1 stated value,...
-
Discuss the fundamental principles of Akhlaq based on Islamic concept? Discuss the sources of Islamic law where the Desired Akhlaq could be found.
-
A couple want to retire together 1 5 years from today. They have managed to save $ 5 0 , 0 0 0 at the bank presently earning 5 . 1 5 % interest per annum, compounding monthly. The couple will also...
-
An analyst with a leading investment bank tracks the stock of Mandalays Inc. According to her estimations, the value of Mandalays Inc. s stock should be $ 2 5 . 0 0 per share, but Mandalays Inc. s...
-
Sarah has a 10, 000 bonus and wantswants to use the funds for investing in publicly traded securities. What will happen if Sarah chooses to place the funds in a brokerage firm compared to a mutual...
-
QUESTION 9 Bank Asset Bond A Bank Liability L Settlement 6/27/2019 Settlement 6/27/2019 Maturity 6/27/2029 Maturity 6/27/2022 Rate 10% Rate 8% Yield 8% Yield 7% Redemption 100 Redemption 100...
-
Study the following Minitab output. What statistical test was being computed by Minitab? What are the results of thisanalysis? Mann-Whitney Test and CI N Median 1st Group 16 37.000 2nd Group 16...
-
A police officer pulls you over and asks to search your vehicle because he suspects you have illegal drugs inside your car. Since he doesn't have reasonable suspicion to search your car, legally he...
-
A sample of Na2SO4(s) is dissolved in 225 g of water at 298 K such that the solution is 0.325 molar in Na 2 SO 4 . A temperature rise of 0.146C is observed. The calorimeter constant is 330. J K 1 ....
-
Assign a name for each of the following compounds. a. b. c.
-
a. Using the relationships derived in Example Problem 7.1 and the values of the critical constants for water from Table 7.2, calculate values for the van der Waals parameters a, b, and R from z c , T...
-
Your manager wants you to help him value the Firm K and its equity and debt. The risk-freerateis 2%whiletheexpected marketreturn is 5%. Firm K's beta is 1.5. Firm's K debt to equity is 1:1. Firm's K...
-
8. Plot the sine curve y = 2 sin( [2x + 3]). Write down the five main points used for graphing. (3 marks) 9. Find two values of x less than 360 such that sin(2x) = 13. ( 3 marks ) - 2 10. Find two...
-
Assume you own a 2 year US Treasury Note with a 2% coupon and a 10 year US Treasury Note with a 1% coupon. If market interest rates increase by 100 basis points in the 2 year maturity and only 50...

Study smarter with the SolutionInn App


