For each of the following electrochemical cells, write equations for the oxidation and reduction half-reactions and for
Question:
For each of the following electrochemical cells, write equations for the oxidation and reduction half-reactions and for the overall reaction.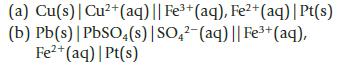
Transcribed Image Text:
(a) Cu(s) | Cu²+ (aq) || Fe³+ (aq), Fe²+ (aq) | Pt(s) (b) Pb(s) | PbSO4(s) | SO4²-(aq) || Fe³+ (aq), Fe²+ (aq) | Pt(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a Cus Cuaq Feaq Feaq Pts Oxidation HalfReaction at the anode Cus Cuaq 2e ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
For each of the following electrochemical cells, write equations for the oxidation and reduction half-reactions and for the overall reaction. (a) Pb(s) | Pb+ (aq) || Sn4+ (aq), Sn+ (aq) | C(s) (b)...
-
Each of the following equations describes a reaction of a compound called methyl formate. To what class of compounds does methyl formate belong? Which reactions require a reducing agent? Which...
-
Write balanced equations for each of the following reactions (some of these are analogous to reactions shown in the chapter). (a) Aluminum metal reacts with acids to form hydrogen gas. (b) Steam...
-
The quarterly sales for a software product over the past three years are given in the table below. 1) Forecast the demand for year 4 using the moving average technique for 3 periods. 1) Compute the...
-
ERP software programs allow tighter linkages within a supply chain than were possible with earlier generations of software. Consider the possibility of a tighter link between the marketing and...
-
What are the three main approaches for preventing fraud discussed in the chapter?
-
Develop the expressions for the mole fractions of reacting species as functions of the reaction coordinate for a system initially containing \(3 \mathrm{~mol} \mathrm{H}_{2} \mathrm{~S}\) and \(5...
-
Aubrae and Tylor Williamson began operations of their furniture repair shop (Furniture Refinishers, Inc.) on January 1, 2019. The annual reporting period ends December 31. The trial balance on...
-
A car is driven east for a distance of 4 0 km , then north for 2 1 km , and then in a direction 2 6 \\ deg east of north for 2 7 km . Determine ( a ) the magnitude of the car\'s total displacement...
-
In Chapter 13, you learned that entropy, as well as enthalpy, plays a role in the solution process. If H for the solution process is zero, explain how the process can be driven by entropy.
-
Chloroacetic acid, ClCH 2 CO 2 H, is a moderately weak acid (K a = 1.40 10 3 ). If you dissolve 94.5 mg of the acid in water to give 125 mL of solution, what is the pH of the solution?
-
Consider a mineral that is in fixed supply, Q S = 4. The demand for the mineral is given by Q D = 10 - 2p, where p is the price per pound and Q D is the quantity demanded. The government imposes a...
-
If you were an economist for a firm that wanted to merge, would you argue that the three-digit or five-digit NAICS industry is the relevant market? Why?
-
Do you believe that women will arrive at different outcomes than men when playing a strategic game? Why? Which is preferable? (Feminist)
-
How do platform businesses change the nature of antitrust policy?
-
What are three reasons fewer antitrust cases have been brought before the courts?
-
In a Vickrey auction how would a persons bid differ if she knew that the seller had someone at the auction submitting a bid for the seller?
-
You are in charge of designing a supply chain for furniture distribution. One of your products is a study desk. This desk comes in two colors: black and cherry. Weekly demand for each desk type is...
-
Explain why it is not wise to accept a null hypothesis.
-
For laser action to occur, the medium used must have at least three energy levels. What must the nature of each of these levels be?
-
If Plancks constant were smaller than it is, would quantum phenomena be more or less conspicuous than they are now?
-
In the Bohr model of the hydrogen atom, the radius of the electrons orbit in the ground state is 5.3 10 -11 m. What aspect of the quantum-mechanical model of this atom would you expect to correspond...
-
Duval Company issues four-year bonds with a $117,000 par value on January 1, 2021, at a price of $112,870. The annual contract rate is 9%, and interest is paid semiannually on June 30 and December...
-
You have just been hired as a financial analyst for Lydex Company, a manufacturer of safety helmets. Your boss has asked you to perform a comprehensive analysis of the company s financial...
-
Identify and describe the basic and precision cuts used in commercial kitchen?

Study smarter with the SolutionInn App


