In Figure 1.5 you see macroscopic and particulate views of the element bromine. Which are the macroscopic
Question:
In Figure 1.5 you see macroscopic and particulate views of the element bromine. Which are the macroscopic views and which are the particulate views? Describe how the particulate views explain properties of this element related to the state of matter.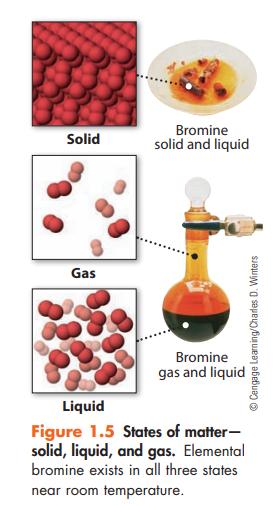
Transcribed Image Text:
Solid Gas Bromine solid and liquid Bromine gas and liquid Liquid Figure 1.5 States of matter- solid, liquid, and gas. Elemental bromine exists in all three states near room temperature. ⒸCengage Learning/Charles D. Winters
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
The macroscopic views of bromine are the images of the solid liquid and gas states of bromine These images show what we can see with our naked eyes Th...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
In Figure 1.2 you see a piece of salt and a representation of its internal structure. Which is the macroscopic view and which is the particulate view? How are the macroscopic and particulate views...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Solve the following system. 10. q = 1 (mod 3) q = 2 (mod 4) q = -5 (mod 5) q = 5 (mod 7) 0
-
Write structures for the following bicyclic alkanes: (a) Bicyclo [1.1.0] butane (b) Bicyclo [2.1.0] pentane (c) 2-Chlorobicyclo [3.2.0] heptane (d) 7-Methylbicyclo [2.2.1] heptane
-
Jacobs Company replaced carpeting throughout its general offices. The old carpet was removed at a cost of $1,500 on March 15. The book value of the old carpet was $6,000 on March 15 ($18,000 original...
-
As HR director for an oil company, you are responsible for selecting 15 expatriates to go to work in Iraq. However, you are personally concerned about the safety there. How do you proceed?
-
A person is required to toss 8 unbiased coins and note down the outcome of each. The random variable represents the number of heads. Determine whether the experiment is a binomial experiment. If it...
-
The Nelson Company bought inventory for $50,000 on terms of 2/15, n/60. It pays for the first $37,500 of inventory purchased within the discount period and pays for the remaining $12,500 two months...
-
Distribution is a statistical concept used in data research. Those seeking to identify the outcomes and probabilities of a particular study will chart measurable data points from a data set,...
-
Identify the following as either physical changes or chemical changes. (a). The desalination of sea water (separation of pure water from dissolved salts). (b). The formation of SO 2 (an air...
-
Identify the following as either physical changes or chemical changes. (a). Dry ice (solid CO 2 ) sublimes (converts directly from solid to gaseous CO 2 ). (b). Mercurys density decreases as the...
-
Why do Starbucks customers at busy downtown locations in major cities pay more for a cup of coffee than they would at less busy locations? a) Starbucks coffee is better than that of any other...
-
Naqvi eBikes sells to three retail dealers, each of whom pays Naqvi monthly. On average, the three dealers pay monthly payments of $132,700, $156,200, and $81,600 respectively. Each payment has a...
-
Approximate the value of the given integral by completing parts (a) and (b) below. (a) Use Simpson's rule, using n = 2. (b) Check by direct integration. 2 (6+x) dx 0
-
Member AB has the angular velocity WAB = 2 rad/s and angular acceleration OAB = 5 rad/s. 100 mm Part B aCB D 11 60 450 mm vec AB Determine the angular acceleration of member CB measured...
-
Amy Weir had the following transactions during the 2021/22 tax year: Assessable business income $ 97,OOO Maintenance receivedfrom ex-spouse ...
-
How do molecular dynamics simulations elucidate the complex interplay between thermodynamic forces and surface interactions in the development of novel nanoparticle catalysts for selective...
-
Sellers Construction Company purchased a compressor for $28,000 cash. It had an estimated useful life of four years and a $4,000 salvage value . At the beginning of the third year of use, the company...
-
Use the T account for Cash below to record the portion of each of the following transactions, if any that affect cash. How do these transactions affect the companys liquidity? Jan. 2 Provided...
-
Assign IUPAC names for each of the following compounds: (a) (b) (c) (d) . CH CH3
-
Assign IUPAC names for each of the following compounds: (a) (b) (c) (d) H H- CH- 13
-
Identify the compounds below that can function as Lewis bases: N'
-
1. The scores of the students in the final examination are normally distributed with a mean of 72 and a standard deviation of 8. a. Find the percentage of students who scored less than 60. b. Find...
-
A subgroup H of S, is called transitive if the standard action by H on {1,..., n} is transitive. Show that the transitive subgroups of S3 are A3 and S3.
-
Chris Smith - Owner and Senior Accountant B.Ec. (Accounting major), M.Tax (UNSW), FIPA Director Chris is a registered tax agent and an accountant who has been in public practice as an accountant...

Study smarter with the SolutionInn App


