Phosgene, Cl 2 CO, is a highly toxic gas that was used as a weapon in World
Question:
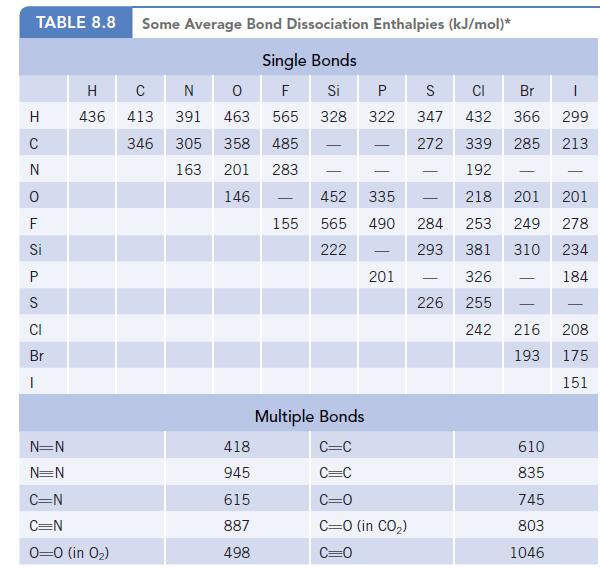
Phosgene, Cl2CO, is a highly toxic gas that was used as a weapon in World War I. Using the bond dissociation enthalpies in Table 8.8, estimate the enthalpy change for the reaction of carbon monoxide and chlorine to produce phosgene.![]()
Data given in table 8.8
Transcribed Image Text:
CO(g) + Cl₂(g) → Cl₂CO(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
CO Cl CICO AHrxn AHbonds brokenAHbonds ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Phosgene is highly toxic and was used as a chemical weapon in World War I. It is also a synthetic precursor used in the production of many plastics. (a) When vapors of phosgene are inhaled, the...
-
Use the bond dissociation enthalpies in Table 8.8 to estimate the enthalpy change for the decomposition of urea to hydrazine, H 2 NNH 2 , and carbon monoxide. (Assume all compounds are in the gas...
-
Phosgene (CC1 2 O) is a colorless gas that was used as an agent of chemical warfare in World War I. It has the odor of newly mown hay (which is a good warning if you know the smell of newly mown...
-
Design a suitable angle section to carry a factored tensile force 210 kN assuming a single row of M20 bolts. The yield strength and ultimate strength of the material is 250 MPa and 410 MPa,...
-
Web services have been called a second wave of net-centric computing that will have broad implications for software development approaches in the future. Develop an argument to support or refute this...
-
The final chapter in your book is dedicated to building high level leadership skills for optimizing results under changing situations. One of these skills is managing conflict. Conflict is always...
-
The Haaland formula for the friction factor is \[ f=\frac{0.3086}{\left\{\log \left[6.9 / \operatorname{Re}+(\varepsilon / 3.7 D)^{1.11} ight] ight\}^{2}} \] Compare this equation for \(f\) for...
-
Transpiration cooling in a planar system, two large flat porous horizontal plates is separated by a relatively small distance L. The upper plate at y = L is at temperature TL, and the lower one at y...
-
The distance (in feet) of an object som a point is given by s(t) = r, where timer is in seconds. (a) What is the average velocity of the object between t = 3 and t = 5? The average velocity between t...
-
Consider the carbonoxygen bond in formaldehyde (CH 2 O) and carbon monoxide (CO). In which molecule is the CO bond shorter? In which molecule is the CO bond stronger?
-
Methanol can be made by partial oxidation of methane using O 2 in the presence of a catalyst: Use bond dissociation enthalpies to estimate the enthalpy change for this reaction. Compare the value...
-
Suppose employment websites develop new software that enables job-seekers to find suitable jobs more quickly and employers to better screen potential employees. What effect will this have on the...
-
On 12 November 2011, Hazel made a gross chargeable transfer to a relevant property trust of 266,000 (after deduction of exemptions). On 1 April 2017 she gave 300,000 to her grandson. These were her...
-
U Ltd has three subsidiaries (one of which is dormant) and has the following results for the year to 31 December 2021: Compute the corporation tax liability for the year and state the date (or dates)...
-
Tracey is a sole trader. She has the following transactions during the quarter to 31 December 2021 (all amounts shown are VAT-exclusive): Tracey drives a car with an emission rating of 128 g/km and...
-
During the quarter to 31 December 2021, a taxable person makes the following supplies: Input tax for the quarter is 118,000. Of this input tax, 35% is attributed to taxable supplies, 40% is...
-
Susan made the following purchases of ordinary shares in Semicycle plc: In January 2022, the company went into liquidation and Susan received a first distribution of 2 per share. The market value of...
-
Write a paper describing in detail how the income statement, statement of owners equity, and balance sheet are related.
-
Smthe Co. makes furniture. The following data are taken from its production plans for the year. Required: 1. Determine the hazardous waste disposal cost per unit for chairs and for tables if costs...
-
Semiconductors can become conductive if their temperature is raised sufficiently to populate the (empty) conduction band from the highest filled levels in the valence band. The ratio of the...
-
For the Ï-network of β-carotene modeled using the particle in the box, the position-dependent probability density of finding 1 of the 22 electrons is given by The quantum number n in...
-
Calculate the energy levels of the -network in hexatriene, C 6 H 8 , using the particle in the box model. To calculate the box length, assume that the molecule is linear and use the values 135 and...
-
Design the Graphical user interface as shown in figure below Measurement: 3.4 Conversion: KM to M Calculate O 3.4 KM in miles is 2.108 OK X Using do while loop compute and display conversion for...
-
Woodbridge Furniture Company produces custom furniture for walk - in customers. Products include coffee tables, shelving units, bookshelf, and hutches and can be constructed with either pine or oak....
-
Trey Monson starts a merchandising business on December 1 and enters into the following three inventory purchases. Monson uses a periodic inventory system. Also, on December 15, Monson sells 27 units...

Study smarter with the SolutionInn App


