The formation of diamond from graphite is a process of considerable importance. (a) Using data in Appendix
Question:
The formation of diamond from graphite is a process of considerable importance.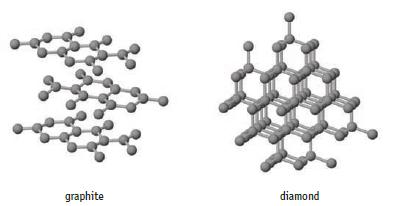
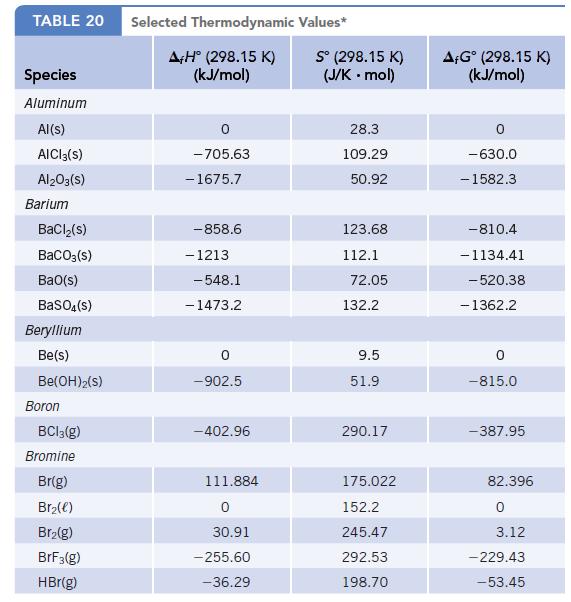
(a) Using data in Appendix L, calculate Δr S°, Δr H°, and ΔrG° for this process at 25°C.
(b) The calculations will suggest that this process is not possible at any temperature. However, the synthesis of diamonds by this reaction is a commercial process. How can this contradiction be rationalized? (Note: In the industrial synthesis, high pressure and high temperatures are used.)
Data given in Appendix L
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:





