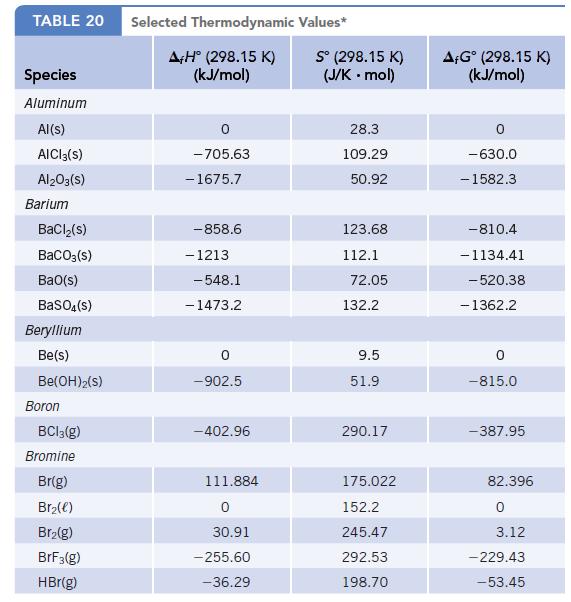
Use values of f G for solid and gaseous iodine at 25C (Appendix L) to calculate
Question:
Use values of Δf G° for solid and gaseous iodine at 25°C (Appendix L) to calculate the equilibrium vapor pressure of iodine at this temperature.
Data given in Appendix L
Transcribed Image Text:
ⒸCengage Learning/Chades D. Winters
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
120 128 AG for 12 000 KJmol AG for I 193 KJmol AG 1x0 193KJmol000 ...View the full answer

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
dC The concentration of a solute at time t is given by C(t) where = 2(20 - C(t)) for t20. dt (a) Solve the differential equation when C(0) = 7. (b) Find limC(t). (c) Use the answer to part (a) to...
-
Use data from Appendix C to calculate the equilibrium constant, K, at 298 K for each of the following reactions: H2(g) + 12(g) 2 HI(g) C2H5OH (g)- C2H4(g) + H2O(g)
-
The following data are the equilibrium vapor pressure of limonene, C 10 H 16 , at various temperatures. (Limonene is used as a scent in commercial products.) (a) Plot these data as ln P versus 1/T so...
-
What does a SWOT analysis reveal about the overall attractiveness of lululemon's situation?
-
In what ways are team cohesiveness and team conflict related?
-
Using the same scenario as question 6, which timing schedule would you choose to use for each communication? Why? Question 6, Imagine you are the project manager of a team tasked with building a new...
-
1. Identify 3 students to play the roles of the employees. Ask these 3 individuals to read their roles below. 2. Identify 1 student to play the role of the president of the social enterprise (Taylor...
-
BizCon, a consulting firm, has just completed its first year of operations. The company's sales growth was explosive. To encourage clients to hire its services BizCon offered 180-day...
-
What is one of the Oracle database upgrade methods. Which of the methods offers the least control over the upgrade process? Please explain thoroughly because I am having a hard time with this subject...
-
The formation of diamond from graphite is a process of considerable importance. (a) Using data in Appendix L, calculate r S, r H, and r G for this process at 25C. (b) The calculations will suggest...
-
Oxygen dissolved in water can cause corrosion in hot-water heating systems. To remove oxygen, hydrazine (N 2 H 4 ) is often added. Hydrazine reacts with dissolved O 2 to form water and N 2 . (a)...
-
Use the phase diagram in Fig. 5.3 to state (i) the solubility of Ag in Sn at 800 C and (ii) the solubility of Ag 3 Sn in Ag at 460 C, (iii) the solubility of Ag 3 Sn in Ag at 300 C. Data in Fig. 5.3...
-
Brad and Angelina are two team leads in your organization. They met working at your company and have started a romantic relationship. They have been seeing each other for over a year. Their work...
-
type out an example email to this hypothetical scenario: A customer messages you distraught that our program is failing him and he is having a horrible day because his dog passed away. He then sends...
-
A company with 20 workers is examining its payroll. The salaries of the workers in thousands are included in the table below. Salaries in Thousands 99 62 2258 57 88 91 16. 75 50 44 Help Copy to...
-
1. You (many years ago) are deciding whether to upgrade your company's equipment from typewriters to computer technology at a cost of $2,500 per machine, and machines will be replaced one-to-one. The...
-
You are the store manager at XYZ wireless. Your duty is to manage the store and the sales team. You get involved in selling only when needed. One day, a customer comes into your store and is greeted...
-
1. Use the background information to create a business profile for New Century, and indicate areas where more information will be needed. The profile should include an organization chart of the...
-
Choose two matrices A and B with dimension 2 x 2. Calculate det A, det B, and det (AB). Repeat this process until you are able to discover how these three determinants are related. Summarize your...
-
Determine the moments at D and C, then draw the moment diagram for each member of the frame. Assume the supports at A and B are pins. EI is constant. 16 kN 3 m D 4 m
-
Determine the moments at B and C, then draw the moment diagram for each member of the frame. Assume the supports at A,E, and D are fixed.EI is constant. 10 k 2 k/ft 8 ft -8 ft PA 12 ft 16 ft
-
The frame is made from pipe that is fixed connected. If it supports the loading shown, determine the moments developed at each of the joints. EI is constant. 18 kN 18 kN 4 m 4 m 4 m
-
It was the first pregnancy for Barbara Ann, 29 years old, and it was normal. During pregnancy, she reported a "small" breast enlargement. Your child is breastfed for the first time within 2 hours...
-
Justin is an unlucky soul who got Herpes Simplex Virus 1 (HSV-1) from his girlfriend in junior high. Now he is a carrier for HSV-1 and anytime he experiences high levels of stress, he develops cold...
-
This is a use case for an UBER like application called 'Mishwar' where a UML design class diagram DCD is required for this use case.Cheshire Cat then made additional payments as follows, based upon...

Study smarter with the SolutionInn App


