The halogens form three stable, weak acids, HOX. (a) Which is the strongest of these acids? (b)
Question:
The halogens form three stable, weak acids, HOX.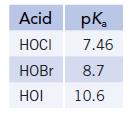
(a) Which is the strongest of these acids?
(b) Explain why the acid strength changes as the halogen atom is changed.
Transcribed Image Text:
Acid pK₂ HOCI 7.46 HOBr 8.7 HOI 10.6
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
a Strongest Acid HOCl Hypochlorous Acid The strength of an acid is determined by its acid dissociati...View the full answer

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
The following information was extracted from Citigroup, Inc.'s 2009 annual report. From letter to shareholders: Financial Strength While Citi started the year as a TARP institution receiving...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
A database is to be made to store information about a catalogue of CDs. Information to be stored about each CD includes title, price, genre, and a list of tracks. Each CD will also have an artist,...
-
1. Do you think that TI took the right approach to achieving better customer satisfaction by training its executives first? Would TI have achieved quicker results by training its front-line employees...
-
Refer to the facts in problem P9-38. Data from P9-38 Due to concerns with climate change resulting from rising levels of carbon dioxide and other greenhouse gases, many companies and organizations...
-
Dry, compressed air at \(T_{m, i}=75^{\circ} \mathrm{C}, p=10 \mathrm{~atm}\), with a mass flow rate of \(\dot{m}=0.001 \mathrm{~kg} / \mathrm{s}\), enters a \(30-\mathrm{mm}-\) diameter, 5 -m-long...
-
(a) From what sources might a corporation obtain funds through long-term debt? (b) What is a bond indenture? What does it contain? (c) What is a mortgage?
-
In the ratification process, there were two sides; what was each side called? How many states had to ratify the new constitution in order to pass it? What proved to be a big problem for the...
-
Perchloric acid behaves as an acid, even when it is dissolved in sulfuric acid. (a) Write a balanced equation showing how perchloric acid can transfer a proton to sulfuric acid. (b) Draw a Lewis...
-
The acidity of the oxoacids was described in Section 16.9, and a larger number of acids are listed in the table below. (a) What general trends do you see in these data? (b) What has a greater effect...
-
The current I (in A) passing through a resistor R (in ) in which P watts of power are dissipated is I = P/R. If the power dissipated in the resistors shown in Fig. 11.7 is W watts, what is the sum of...
-
What guidelines should be followed when planning and incorporating visuals into a presentation?
-
Write an email to your instructor summarizing your progress on your report. In the introductory paragraph, summarize your progress in terms of your schedule and your goals. Under the heading Work...
-
What are three guidelines for delivering in-person presentations and three guidelines for delivering virtual presentations?
-
What are some things a person can do to mitigate fear when preparing and delivering an oral presentation?
-
What is the role of networking and internships when you are looking for a job?
-
San Tan Company manufactures light bulbs. Once produced, the bulbs are packaged and sold to wholesalers for distribution to grocery stores and large retailers like Walmart. The following costs were...
-
Suppose a population of bacteria doubles every hour, but that 1.0 x 106 individuals are removed before reproduction to be converted into valuable biological by-products. Suppose the population begins...
-
What is the definition of zero energy employed in constructing the statistical mechanical expression for the equilibrium constant? Why was this definition necessary?
-
Why should the equilibrium constant be dependent on the difference in Gibbs energy? How is this relationships described using statistical mechanics?
-
For the equilibrium involving the dissociation of a diatomic, what energetic degrees of freedom were considered for the diatomic and for the atomic constituents?
-
Find the logarithm using the change of base formula. log 30.13 log 30.13= (Round to four decimal places as needed.)
-
Find the least common multiple of these two expressions. 5 10x us and 25x u y7
-
Solve the following system of equations for z and for y: System of Equations: y=9+3z y = 51 - 3z Value of z Value of y Solve the following system of equations for a and for b: System of Equations:...

Study smarter with the SolutionInn App


