Use standard reduction potentials (Appendix M) for the half-reactions AgBr(s) + e Ag(s) + Br
Question:
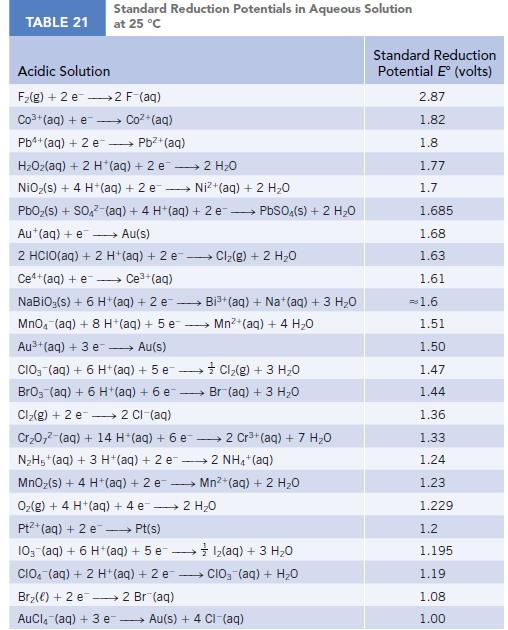
Use standard reduction potentials (Appendix M) for the half-reactions AgBr(s) + e− → Ag(s) + Br−(aq) and Ag+(aq) + e− → Ag(s) to calculate the value of Ksp for AgBr.
Data given in Appendix M
Transcribed Image Text:
TABLE 21 Standard Reduction Potentials in Aqueous Solution at 25 °C Acidic Solution F₂(g) + 2 e 2 F-(aq) Co3+ (aq) + e Coz+(aq) Pb4+ (aq) + 2 e - Pb²+ (aq) HzOz(aq) + 2 H*(aq) +2e → 2H2O NiO₂(s) + 4 H+ (aq) + 2 e→→→→→→ Ni+(aq) + 2 HO PbO₂ (s) + SO4² (aq) + 4 H+ (aq) + 2e → PbSO4(s) + 2 H₂O Au+ (aq) + e→→→→→ Au(s) 2 HCIO(aq) + 2 H+ (aq) + 2 e-- - Ce+(aq) + e→→→→ Ce³+ (aq) NaBiO;(s) + 6 H+ (aq) + 2 e- → MnO4 (aq) + 8 H+ (aq) + 5 e Au³+ (aq) + 3 e→→→→→ Au(s) CIO3(aq) + 6 H+ (aq) + 5 e→→→→→→ BrO3 (aq) + 6 H+ (aq) + 6 e- Cl₂(g) + 2 e 2 Cl-(aq) Cr₂0,² (aq) + 14 H*(aq) + 6 e 2 Cr³+ (aq) + 7 H₂O N₂H5+ (aq) + 3 H+ (aq) + 2 e2 NH4+ (aq) MnO₂ (s) + 4 H+ (aq) + 2 e O₂(g) + 4 H+ (aq) + 4 e 2 H₂O Pt+ (aq) + 2 e →→→→→ Pt(s) 10- (aq) + 6 H+ (aq) + 5 e1₂(aq) + 3 H₂O CIO (aq) + 2 H+ (aq) + 2 e CIO₂ (aq) + H₂O Br₂() +2 e 2 Br(aq) AuCl(aq) + 3 e → Cl₂(g) + 2 H₂O → Bi³+ (aq) + Na+ (aq) + 3 H₂O Mn²+ (aq) + 4H₂O Cl₂(g) + 3 H₂O Br (aq) + 3 H₂O Mn²+ (aq) + 2 H₂O Au(s) + 4 CI-(aq) Standard Reduction Potential E (volts) 2.87 1.82 1.8 1.77 1.7 1.685 1.68 1.63 1.61 = 1.6 1.51 1.50 1.47 1.44 1.36 1.33 1.24 1.23 1.229 1.2 1.195 1.19 1.08 1.00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
2303 RT E log Ksp nF nFE log Ksp 2303 RT 1x9...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The standard reduction potentials of the following half-reactions are given in Appendix E: (a) Determine which combination of these half-cell reactions leads to the cell reaction with the largest...
-
Use the standard reduction potentials (Appendix M) for the half-reactions [Zn(OH) 4 ] 2 (aq) + 2 e Zn (s) + 4 OH (aq) and Zn 2+ (aq) + 2e Zn(s) to calculate the value of K formation for the...
-
Use the table of standard reduction potentials (Appendix M) to calculate r G for the following reactions at 298 K. Data given in Appendix M (a) CIO3(aq) + 5 Cl(aq) + 6 H+ (aq) 3 Cl(g) + 3 HO(l) (b)...
-
A rectangular pontoon 10m long 7m broad & 2.5m deep weights 686.7KN. It carries on its upper deck an empty boiler of 5m diameter weighing 588.6 KN. The centre of gravity of the boiler and pontoon are...
-
Koko Chocolate Company makes dark chocolate and light chocolate. Both products require cocoa and sugar. The following planning information has been made available: Koko Chocolate does not expect...
-
Excel Project: Fixing Cryptic Data using Data Concatenation You work at a non-profit that seeks to help people start businesses in third world countries so they can lift themselves out of poverty....
-
Discuss briefly the five areas of project feasibility.
-
During its first year of operations, Gehrig Company had credit sales of $3,000,000, of which $400,000 remained uncollected at year-end. The credit manager estimates that $18,000 of these receivables...
-
Mike is the Controller at Combine Supply Company. Combine Supply has an investment account with a large brokerage. Mike is authorized to call the broker and initiate trades on behalf of the company....
-
Diagram the apparatus used to electrolyze molten NaCl. Identify the anode and the cathode. Trace the movement of electrons through the external circuit and the movement of ions in the electrolysis...
-
Calculate r G and the equilibrium constant for the following reactions. (a) Zn+ (aq) + Ni(s) Zn(s) + Ni+ (aq) (b) Cu(s) + 2 Ag+ (aq) = Cu+ (aq) + 2 Ag(s)
-
Mention the different methods of by-product cost accounting.
-
Suppose that Congress repeals MLB's exemption from the antitrust laws. How might this affect the contract zone between MLB and the MLBPA?
-
Explain how each of the following would affect the NHLPA's bargaining position. a. Russia's Kontinental Hockey League signs a large number of NHL stars. b. A change in the tax laws increases the...
-
How might the development of the NFL and MLB have been different if blacks had not been excluded from these leagues during the years that spanned World War II?
-
Think of recent events from the sports industry that show a taste for discrimination. Do they represent consumer, employer, or employee discrimination?
-
You have been asked ro design a series of user interface screens that will be used by both employees and customers. You proudly roll out your prototypes, expecting accolades and high pratse. Instead,...
-
A company manufactures a liquid product called Crystal. The basic ingredients are put into process in Department 1. In Department 2, other materials are added that increase the number of units being...
-
Question 6.10 Current and deferred tax worksheets and tax entries From the hip Ltd?s statement of profit or loss for the year ended 30 June 2007 and extracts from its statements of financial position...
-
A hockey puck slides along a rough, icy surface. It has an initial velocity of 35 m/s and slides to a stop after traveling a distance of 95 m. Find the coefficient of kinetic friction between the...
-
A rock is dropped from a very tall tower. If it takes 4.5 s for the rock to reach the ground, what is the height of the tower?
-
A baseball is hit directly upward with an initial speed of 45 m/s. Find the velocity of the ball when it is at a height of 40 m. Is there one correct answer for v or two? Explain why.
-
In January, Tongo, Incorporated, a branding consultant, had the following transactions. Indicate the accounts, amounts, and direction of the effects on the accounting equation under the accrual...
-
Explain the design principles behind file systems, including the implementation of hierarchical directory structures, metadata management, and file allocation strategies .
-
On March 1 of the current year, Spicer Corporation compiled Information to prepare a cash budget for March, April, and May. All of the company's sales are made on account. The following Information...

Study smarter with the SolutionInn App


