Using Figure 17.11, suggest an indicator to use in each of the following titrations. (a) NaHCO 3
Question:
Using Figure 17.11, suggest an indicator to use in each of the following titrations.
(a) NaHCO3 is titrated to CO32− with NaOH.
(b) Hypochlorous acid is titrated with NaOH.
(c) Trimethylamine is titrated with HCl.
Data given in Figure 17.11
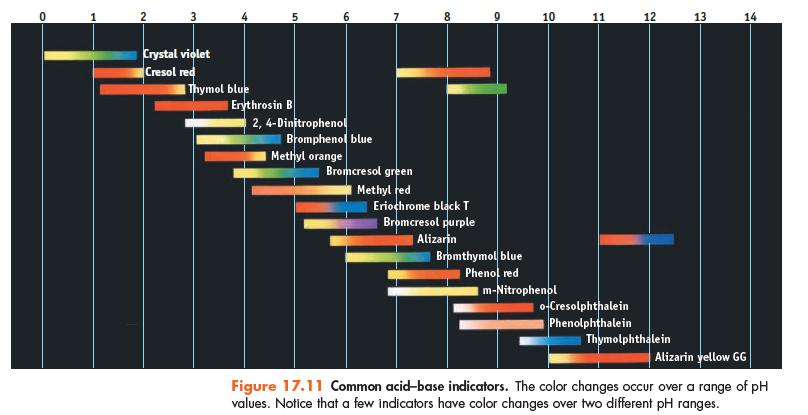
Transcribed Image Text:
3 Crystal violet Cresol red Thymol blue 5 Erythrosin B 2,4-Dinitrophenol Bromphenol blue Methyl orange 7 Bromcresol green Methyl red 00 Eriochrome black T Bromcresol purple Alizarin 8 10 Bromthymol blue Phenol red Im-Nitrophenol 11 o-Cresolphthalein Phenolphthalein 12 Thymolphthalein 13 14 Alizarin yellow GG Figure 17.11 Common acid-base indicators. The color changes occur over a range of pH values. Notice that a few indicators have color changes over two different pH ranges.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a NaHCO3 is titrated to CO32 with NaOH Phenolphthalein indic...View the full answer

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Using Figure 17.11, suggest an indicator to use in each of the following titrations: (a) The weak base pyridine is titrated with HCl. (b) Formic acid is titrated with NaOH. (c) Ethylenediamine, a...
-
Suggest a suitable indicator for a titration based on each of the following reactions. Use 0.05 M if an equivalence point concentration is needed. *(a) H2CO3 + NaOH NaHCO3 + H2O (b) H2P + 2NaOH ...
-
Del Gato Clinic's cash account shows an $14,180 debit balance and its bank statement shows $13,236 on deposit at the close of business on June 30. a. Outstanding checks as of June 30 total $1,502. b....
-
Carl contributes equipment with a $50,000 adjusted basis and an $80,000 FMV to Cook Corporation for 50 of its 100 shares of stock. His son, Carl Jr., contributes $20,000 cash for the remaining 50...
-
What are some reasons for the implementation of ESOPs? Cite some of the potential problems concerning their use.
-
Chicago-based mail-order supplier of sailboat hardware and equipment. In the mid-1970s, Comer began to emphasize clothing and soft luggage and subsequently eliminated sailboat hardware from the...
-
An electronic instrument is to be isolated from a panel that vibrates at frequencies ranging from \(25 \mathrm{~Hz}\) to \(35 \mathrm{~Hz}\). It is estimated that at least 80 percent vibration...
-
A group of medical professionals is considering the construction of a private clinic. If the medical demand is high (i.e., there is a favorable market for the clinic), the physicians could realize a...
-
A 1000 gallon holding tank that catches runoff from some chemical process initially has 800 gallons of water with 2 ounces of pollution dissolved in it. Polluted water flows into the tank at a rate...
-
Name two insoluble salts of each of the following ions. (a) Cl (b) Zn 2+ (c) Fe 2+
-
Construct a rough plot of pH versus volume of base for the titration of 25.0 mL of 0.050 M HCN with 0.075 M NaOH. (a) What is the pH before any NaOH is added? (b) What is the pH at the halfway point...
-
The following direct materials and direct labor data pertain to the operations of Engles Manufacturing Company for the month of August. Instructions (a) Compute the total, price, and quantity...
-
What is a personality trait you have in common with your parents? Perhaps it is a reaction you have in certain situations or something you find yourself saying. Is it a good or bad thing you picked...
-
Discuss how men and women differ in their efforts to end their lives. How do marriage, age, and ethnicity influence these differences? 3. Discuss suicide prevention strategies.
-
Reference the article on Workplace Wellness Programs Workplace Wellness Programs - Alternative Formats , which provides an example of a complete policy. Compile a concise model policyon opioid...
-
Newton's Universal Law of Gravity (NULG) A) Discuss Newton's Universal Law of Gravity (NULG). a. What is required for there to be gravity? b. On what does gravity depend? c. What is the difference,...
-
How you and management at your organization could reduce the potential impact of a strike? Explain in details.
-
Which indexes can you use to compare your investment performance to general market returns? Briefly explain each of these indexes.
-
Define a traverse in Surveying?
-
A crude model for the molecular distribution of atmospheric gases above the Earths surface (denoted by height, h) can be obtained by considering the potential energy due to gravity: In this...
-
Another use of distribution functions is determining the most-probable value, which is done by realizing that at the distribution maximum the derivative of the distribution function with respect to...
-
In nonlinear optical switching devices based on dye-doped polymer systems, the spatial orientation of the dye molecules in the polymer is an important parameter. These devices are generally...
-
1. Subtract B from A, then display the difference. 2. Convert the following LMC Mnemonic code Machine code 00 IN 01 STD 99 02 IN 03 ADD 99 04 STD 99 05 IN 06 STD 98 07 LDA 99 08 SUB 98 09 OUT 10 COB...
-
A 80kg astronaut is training in human centrifuge to prepare for a launch. The astronaut uses the centrifuge to practice having a 5.51g force (5.51 times their own weight) on their back. Number of...
-
2. From the solid model presented in the following figure, (a) find the coordinates of the center C stored in the model database and the respective mapping matrix [T] that maps point from WCS for the...

Study smarter with the SolutionInn App


