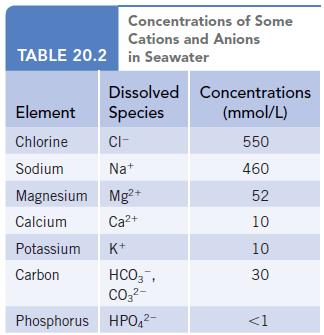
Using the data in Table 20.2, estimate the minimum mass of solid that would remain after evaporation
Question:
Using the data in Table 20.2, estimate the minimum mass of solid that would remain after evaporation of 1.0 L of seawater.
Data given in Table 20.2
Transcribed Image Text:
Concentrations of Some Cations and Anions TABLE 20.2 in Seawater Dissolved Concentrations Species (mmol/L) 550 460 52 10 10 Element Chlorine CI- Sodium Na+ Magnesium Mg²+ Calcium Ca²+ Potassium K+ Carbon HCO3, CO3²- Phosphorus HPO4²- 30 <1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Dissolved Species Element Concentrations mmolL Chloride Cl 550 Sodium Na 460 Magnesium Mg2 52 Calciu...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The Tastee Bakery Company supplies a bakery product to many supermarkets in a metropolitan area. The company wishes to study the effect of the height of the shelf display employed by the supermarkets...
-
In Chapters 2 (Section 2.8) and 3 (Section 3.7) we analyzed the risk and return of the Orange County portfolio, using the building blocks of zero coupon bonds and duration in this exercise, we...
-
Using the data in Table 21.1, compare the price on July 24, 2009, of the following options on JetBlue stock to the price predicted by the Black-Scholes formula. Assume that the standard deviation of...
-
Evaluate the expression for the given value(s) of the variable(s). 4x - 7, x = 5/2
-
Ross-Simmons sued Weyerhaeuser for antitrust violation for driving it out of business. The claim was that Weyerhaeuser consistently outbid Ross for logs to process into lumber. Ross contended this...
-
Why is the greenhouse effect a good thing for life on Earth? Why is it a bad thing?
-
The fugacity co-efficient of a substance is the ratio of its fugacity to the (a) Mole fraction (c) Pressure (b) Activity (d) Activity co-efficient.
-
Barry Bird opened the Barry Bird Basketball Camp for children ages 10 through 18. Campers typically register for one week in June or July, arriving on Sunday and returning home the following...
-
Describe three behaviors Theranos, CEO Elizabeth Holmes displayed, and how they contributed to toxicity. https://youtu.be/jIqF95qiQSs
-
In any solution, there must be a balance between the positive and negative charges of the ions that are present. Use the data in Table 20.2 to determine how close to a balance of charge is achieved...
-
Which of the following statements is not correct? (a) Phosphates enhance the growth of algae in surface water. (b) Melting of the polar ice cap will result in a rise in sea levels. (c) Heavy metal...
-
Washington Corporation has 1,000,000 shares of common stock issued and outstanding with a $ 1 par value. If Washington declares a one- for- four reverse stock split, how many shares will be issued...
-
How does budgeting help the management function of control?
-
What is meant by the present value of a cash flow?
-
Give three reasons for regarding variance reports as a useful tool of management.
-
Explain the meaning of 'non-financial performance measures'.
-
What are the advantages and limitations of the payback method?
-
Use the information in Exercise to prepare an October 31 statement of cash flows for Ernst Consulting. Also assume the following: a. The owners initial investment consists of $38,000 cash and $46,000...
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
If the electric field vector E(z, t) of an EM wave in vacuum is, at a certain location and time, given by vector E = (10 V/m)(cos 0.5) i write an expression for the associated vector B-field.
-
The E-field of an electromagnetic wave is described by Write an expression for the B-field. Determine vector B(0, 0). 3 (i + jEo sin (kz ot + /6)
-
Starting with Eq. (3.32), prove that the energy densities of the electric and magnet fields are equal (u E = u B ) for an electromagnetic wave. (3.32) UB 2o
-
A soap film in air has a thickness of 175 nm. If the index of refraction of the soap film is 1.35, what colour of light will be transmitted through? (i.e., What colour does the bubble appear to be...
-
Two objects, A and B, start from rest. Object A starts with acceleration 4.0 m/s^2 and 3.2 seconds later after A, object B starts in the same direction with acceleration 8.6 m/s^2. What will be speed...
-
A car moving with over-speed limit constant speed 36.7 m/s passes a police car at rest. The police car immediately takes off in pursuit, accelerating with 7.5 m/s^2. How long does it take for police...

Study smarter with the SolutionInn App


