You have a mixture of oxalic acid, H 2 C 2 O 4 , and another solid
Question:
You have a mixture of oxalic acid, H2C2O4, and another solid that does not react with sodium hydroxide. If 29.58 mL of 0.550 M NaOH is required to titrate the oxalic acid in the 4.554-g sample to the second equivalence point, what is the mass percent of oxalic acid in the mixture? Oxalic acid and NaOH react according to the equation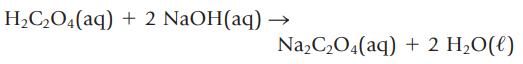
Transcribed Image Text:
H₂C₂O4(aq) + 2 NaOH(aq) Na₂C₂O4(aq) + 2 H₂O(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To find the mass percent of oxalic acid in the mixture you can follow these steps Calculate the mole...View the full answer

Answered By

Munibah Munir
I've done MS specialization in finance’s have command on accounting and financial management. Forecasting and Financial Statement Analysis is basic field of my specialization. On many firms I have done real base projects in financial management field special forecasting. I have served more than 500 Clients for more than 800 business projects, and I have got a very high repute in providing highly professional and quality services.I have capability of performing extra-ordinarily well in limited time and at reasonable fee. My clients are guaranteed full satisfaction and I make things easy for them. I am capable of handling complex issues in the mentioned areas and never let my clients down.
4.60+
467+ Reviews
648+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
You have 0.954 g of an unknown acid, H 2 A, which reacts with NaOH according to the balanced equation If 36.04 mL of 0.509 M NaOH is required to titrate the acid to the second equivalence point, what...
-
A 2.500-g sample of a mixture of sodium carbonate and sodium chloride is dissolved in 25.00 mL of 0.798 M HCl. Some acid remains after the treatment of the sample. a. Write the net ionic equation for...
-
A 2.500-g sample of a mixture of sodium hydrogen carbonate and potassium chloride is dissolved in 25.00 mL of 0.437 M H2SO4. Some acid remains after treatment of the sample. a. Write both the net...
-
} S 1995 the # of Farms Century, data year 1935 1990 19.50 Aumcon people living on declined steadily during the shown by the Follow g as (in milion of persons) from 1935 19.55 1960 1965 11975 1980...
-
A manufacturer of printed circuit boards uses exponential smoothing with trend to forecast monthly demand of its product. At the end of December, the company wishes to forecast sales for January. The...
-
A set of scores measuring aggression is normally distributed with a mean equal to 23 and a standard deviation equal to 2.5. Find the proportion: (a) To the left of x = 19.0 (b) To the right of x =...
-
The Wide World of Fluids article titled "A Sailing Ship without Sails,". Determine the magnitude of the total force developed by the two rotating cylinders on the Flettner "rotor-ship" due to the...
-
Identify which control activity is violated in each of the following situations, and explain how the situation creates an opportunity for fraud or inappropriate accounting practices. 1. Once a month,...
-
1. Discuss one form of radiation you encounter in your daily life and why you use it. 2. What are some of the reported effects of that type of radiation on the human body? [Sources of information...
-
(a) What is the pH of a 0.105 M HCl solution? (b) What is the hydronium ion concentration in a solution with a pH of 2.56? Is the solution acidic or basic? (c) A solution has a pH of 9.67. What is...
-
Sodium thiosulfate, Na 2 S 2 O 3 , is used as a fixer in black-and-white photography. Suppose you have a bottle of sodium thiosulfate and want to determine its purity. The thiosulfate ion can be...
-
Suppose that the life span of a certain automobile tire is normally distributed, with = 25,000 miles and = 2000 miles. (a) Find the probability that a tire will last between 28,000 and 30,000...
-
The company also will increase sales of its cheap clubs by 11,400 sets. The cheap clubs sell for $415 and have variable costs of $145 per set. The fixed costs each year will be $9,300,000. The...
-
Asteroids with a diameter of ~50m are estimated to collide with Earth every ~750 years. Such an event is expected to generate an airburst (atmospheric explosion) with an energy equal to 5.2 Megatons...
-
Project the future revenue growth rate for the next three years of the company. Explain the assumptions you used to predict the company's revenue growth rate(s) for the next three years. Project the...
-
Marie has had a whirlwind of a first semester of college, and now that finals are over, she is concerned about her weight gain during the past few months. After reading a bit about this issue, Marie...
-
Assume that the expected market return is 12% and volatility is 20%. Assume that the CAPM accurately describes the data we are using. the correlation between IBM and the market is 0.90% and the...
-
What does the debt to equity ratio show and how is it calculated?
-
A woman at a point A on the shore of a circular lake with radius 2 mi wants to arrive at the point C diametrically opposite on the other side of the lake in the shortest possible A time. She can walk...
-
Identify a systematic (IUPAC) name for each of the following compounds a. b. c. d. (e) CH 3 (CH 2 ) 4 CO 2 H (f) CH 3 (CH 2 ) 3 COCl (g) CH 3 (CH 2 ) 4 CONH 2 O: NH2
-
Identify the common name for each of the following compounds: a. b. c. d.
-
Draw the structures of eight different carboxylic acids with molecular formula C 6 H 12 O 2 . Then, provide a systematic name for each compound, and identify which three isomers exhibit chirality...
-
Do a 5 Year Valuation with 100% capacity (0% Vacancy), Case #1 Pension Funds Name Empire Hotel Location Rochester, NY Acquisition Year 1994 Year of Construction 1969 Cost $ 9,800,000.00 Occupancy 60%...
-
what are some challenges researchers might encounter when conducting experiments to determine causation?
-
An investor web page claims thatearnings per share is the bestmeasure of a share's true price because it shows you how much of a company's profit after tax that each shareholder owns. Look up the...

Study smarter with the SolutionInn App


