Based on Figure 3.1, determine (a) The percentage of sunlight that is captured by photosynthesis, (b) The
Question:
Based on Figure 3.1, determine
(a) The percentage of sunlight that is captured by photosynthesis,
(b) The percentage of photosynthesis that occurs in land plants and the percentage that occurs in aquatic plants, and
(c) The percentage of the energy captured by all plants that is eventually released when the plants decay.
Figure 3.1
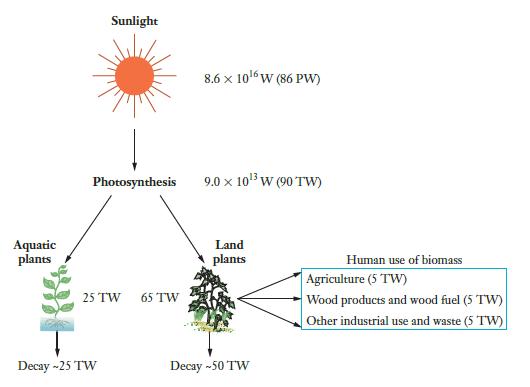
Transcribed Image Text:
Aquatic plants Sunlight Photosynthesis 25 TW Decay - 25 TW 65 TW 8.6 x 106W (86 PW) 9.0 × 10¹³ W (90 TW) Land plants Decay - 50 TW Human use of biomass Agriculture (5 TW) Wood products and wood fuel (5 TW) Other industrial use and waste (5 TW)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
a 010 ...View the full answer

Answered By

Gauri Hendre
I worked as EI educator for Eduphy India YT channel. I gave online tutorials to the students who were living in the villages and wanted to study much more and were preparing for NEET, TET. I gave tutions for topics in Biotechnology. I am currently working as a tutor on course hero for the biochemistry, microbiology, biology, cell biology, genetics subjects. I worked as a project intern in BAIF where did analysis on diseases mainly genetic disorders in the bovine. I worked as a trainee in serum institute of India and Vasantdada sugar institute. I am working as a writer on Quora partner program from 2019. I writing on the topics on social health issues including current COVID-19 pandemic, different concepts in science discipline. I learned foreign languages such as german and french upto A1 level. I attended different conferences in the science discipline and did trainings in cognitive skills and personality development skills from Lila Poonawalla foundation. I have been the member of Lila poonawalla foundation since 2017. Even I acquired the skills like Excel spreadsheet, MS Office, MS Powerpoint and Data entry.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Determine the percentage of sunlight that is absorbed by atmospheric CO2 at a wavelength of = 4.85 m. Assume that the sun is directly overhead, such that the sunlight travels the shortest distance...
-
The sun supplies energy at a rate of about 1.0 kilowatt per square meter of surface area (1 watt = 1 J/s). The plants in an agricultural field produce the equivalent of 20 kg of sucrose (C12H22O11)...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
You put $10,000 in an account earning 5%. After 3 years, you make another deposit into the same account. Four years later (that is, 7 years after your original $10,000 deposit), the account balance...
-
In the year 2008, a real estate analyst forecasts that a rental apartment building will generate $5.3 million each year in rents over the five years 2009-2013. Cash expenses are expected to be $4.2...
-
Defendant Kenneth Blake was married to Charlene Hinton-Blake, who died in 2012. Three of Hinton-Blakes sisters, including Yvonne Hinton, assisted her with day-to-day care during a four-year period of...
-
Biko owns a snowmobile manufacturing business, and Miles owns a mountain bike manufacturing business. Because each business is seasonal, their manufacturing plants are idle during their respective...
-
Let C be the closed convex set shown below u x2 21 Note that C is the intersection of three halfspaces and can be precisely described by the inequalities C = {x R x-1, x20, x1 + x2 0}. (a) Describe...
-
Chlorine has only two isotopes, one with mass 35 and the other with mass 37. One is present at roughly 75% abundance, and the atomic weight of chlorine on a periodic table is 35.45. Which must be the...
-
A solution of ethanol in water has a volume of 54.2 mL and a mass of 49.6 g. What information would you need to look up and how would you determine the percentage of ethanol in this solution?
-
In Exercises 126, graph each inequality. y = log (x + 1)
-
Dozer, Inc. is a closely-held corporation incorporated in a state that has adopted the Model Business Corporation Act. Prior to 2012, Dozers business was organized into two divisions; neither was...
-
3 The label on a company's energy drinks claims that they contain an average caffeine concentration of 250 mg/oz. The mean caf- feine concentration of 15 randomly sampled drinks was 267, with a...
-
You must use the Law of Sines and the Law of Cosines. The air velocity of a jet is 400 km/h [S 30 W]. A 70 km/h wind is blowing from [N20W]. Determine the ground velocity of the jet. Include a proper...
-
My office has two chairs and I like students to be able to sit down when they come to office hours. I don't mind standing. Suppose that 1% of students in the class I teach attends my office hours....
-
A justly-intonated "perfect fifth" in musical harmony is described by a frequency ratio of 3:2. A perfect fifth is a very pleasant pair of notes to hear. An "octave" is a culturally universal musical...
-
You are considering the purchase of an office building for $1.5 million today. Your expectations include the following: first-year potential gross income of $340,000; vacancy and collection losses...
-
Calculate the Lagrange polynomial P 2 (x) for the values (1.00) = 1.0000, (1.02) = 0.9888, (1.04) = 0.9784 of the gamma function [(24) in App. A3.1] and from it approximations of (1.01) and (1.03).
-
Is the ratio of fugacity to pressure greater to or less than one if the attractive part of the interaction potential between gas molecules dominates?
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. HO. HO.
-
A 1.25 mole sample of an ideal gas is expanded from 320. K and an initial pressure of 3.10 bar to a final pressure of 1.00 bar, and C P,m = 5/2R. Calculate w for the following two cases: a. The...
-
4. Define the set inclusion partial ordering on the powersets of a set X. (a) Show that budget correspondence B(p,m) = {x 0|p1x1 + p2x2 m} is monotone increasing in m under set inclusion for fixed p...
-
Remember that the Pumping Lemma is a theorem that holds for every regular language L. It has the quantifier structure, BEL (+). This is equivalent to say that if the corresponding statement with...
-
== This state diagram defines an NFA M ({90, 91, 92), 6, 40, {q0, q1}) accepting some language L over the alphabet = {0, 1}. Step by step follow the algorithm to trans- form this NFA into an...

Study smarter with the SolutionInn App


