Chlorine has only two isotopes, one with mass 35 and the other with mass 37. One is
Question:
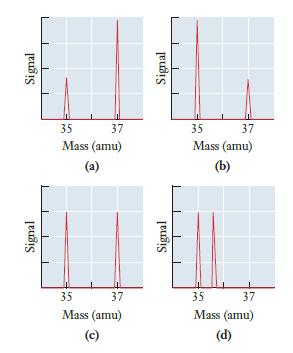
Chlorine has only two isotopes, one with mass 35 and the other with mass 37. One is present at roughly 75% abundance, and the atomic weight of chlorine on a periodic table is 35.45. Which must be the correct mass spectrum for chlorine?
Transcribed Image Text:
Signal Signal 35 37 Mass (amu) (a) 35 37 Mass (amu) (c) Signal Signal T 35 37 Mass (amu) (b) 35 37 Mass (amu) (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
b JE...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
A researcher wanted to find out if there was difference between older movie goers and younger movie goers with respect to their estimates of a successful actors income. The researcher first...
-
A student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all the NBS has reacted. After a careful distillation, the product mixture contains two major...
-
For each case in the accompanying table, answer the questions that follow. a. Calculate the future value of the annuity assuming that it is (1) An ordinary annuity. (2) An annuity due. b. Compare...
-
Why do trailing P/E ratios vary with dividend payout?
-
John and Jennifer Margeson entered into a contract to sell a weight-loss franchise business called Inches-A-Weigh to Theresa Artis. The parties memorialized their agreement in an Asset Purchase...
-
The Kelly Services, Inc., and Subsidiaries balance sheets from its 2008 annual report are presented in Exhibit 5-4. Required a. Using the balance sheets, prepare a vertical common-size analysis for...
-
2. Craig likes to collect records. Last year he had 10 records in his collection. Now he has 14 records. What is the percent increase of his collection?
-
Name each of the following compounds: (a) MgCl 2 , (b) Fe (NO 3 ) 2 , (c) Na 2 SO 4 , (d) Ca (OH) 2 , (e) FeSO 4
-
Based on Figure 3.1, determine (a) The percentage of sunlight that is captured by photosynthesis, (b) The percentage of photosynthesis that occurs in land plants and the percentage that occurs in...
-
What motivates someone to pursue the study of medicine? A students interest and motivation to study medicine can depend on the strength of motivation and career-related values and approaches to...
-
In the United States, the annual salary of someone without a college degree is (on average) $31,752, whereas the annual salary of someone with a college degree is (on average) $49,941. If the cost of...
-
A racing bicycle is travelling on a curve along a track. The curve can be represented as an arc of a circle with a radius of 0.5 km. if the bicycle's speed is 287 km, what is the angle of the curve...
-
The pilot of an aircraft sets the autopilot to level off at 7,000 ft MSL while in a descent. The pilot begins briefing the next phase of flight and when he looks up he notices the altitude of the...
-
Reflect on a significant organizational change that you experienced as a stakeholder. Evaluate that change initiative and the resulting impact to stakeholders. Consider Kotter's eight-step process...
-
For each problem, find the equation of the line tangent to the function at the given point. Your answer should be in slope-intercept form. 7) (x) = x + 2x + 4 at (2, 8) f(x) = x - x +3 at (-1, 1) 9)...
-
An investment opportunity having a market price of $1,000,000 is available. You could obtain a $750,000, 25-year mortgage loan requiring equal monthly payments with interest at 7.0 percent. The...
-
Using the information in P11-2B, compute the overhead controllable variance and the overhead volume variance. Data From Problem 11-2B: Huang Company uses a standard cost accounting system to account...
-
Starting with 1-butanol, show the reagents you would use to prepare each of the following compounds. a. b. c. d. e. H. .
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. .
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. .
-
8. A veterinarian knows that the amount of anesthesia needed to put an animal to sleep for surgery is proportional to the animal's weight. If 2 milliliters of anesthesia will put a 100 pound animal...
-
6. A photograph has a width of 83 cm and an area of 1572 cm. Determine the length of the photograph in the simplest mixed form. (2 marks) 7. Determine the area and perimeter of the square in simplest...
-
46. Approximately 1.5% of the population have a peanut allergy. If East Central Middle School has 1180 students and West Chester Middle School has 1373 students, approximately how many students at...

Study smarter with the SolutionInn App


