Consult a table of standard reduction potentials and determine which of the following reactions are spontaneous under
Question:
Consult a table of standard reduction potentials and determine which of the following reactions are spontaneous under standard electrochemical conditions.
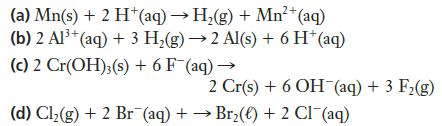
Transcribed Image Text:
2+ (a) Mn(s) + 2 H+ (aq) → H₂(g) + Mn²+ (aq) (b) 2 Al³+ (aq) + 3 H₂(g) → 2 Al(s) + 6 H* (aq) (c) 2 Cr(OH)3(s) + 6 F (aq) → 2 Cr(s) + 6 OH(aq) + 3 F₂(g) (d) Cl₂(g) + 2 Br (aq) + →Br₂() + 2 Cl¯(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a Spontaneou...View the full answer

Answered By

Biplab Sutradhar
I'm a Master's in Computer Application with 10 years of experience in the Information Technology Industry and 5 years of experience as a home tutor. As a tutor, I have extensive hands-on experience in providing individualized instruction to students in a variety of subject areas. I am highly proficient in breaking down complex topics into smaller, more manageable pieces, and I have a strong ability to explain concepts in a clear and concise way. I am experienced in creating engaging learning activities and finding innovative ways to keep students engaged and motivated. I also have experience in developing individualized learning plans to meet the specific needs of each student. Additionally, I am an expert in providing feedback and guidance to help students reach their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Suppose that you work for a company that designs the drive mechanisms for large ships. The materials in this mechanism will obviously come into contact with environments that enhance corrosion. To...
-
1. _____ is the process, procedures, and steps used to make sure digital certificates are up to date and stored securely. A. CSR B. CRL C. Key management D. CN 2. _____ is a type of...
-
Use the table of standard reduction potentials (Appendix M) to calculate r G for the following reactions at 298 K. Data given in Appendix M (a) 3 Cu(s) + 2NO3(aq) + 8 H+ (aq) 3 Cu+ (aq) + 2 NO(g) +...
-
In the dynamic and expanding urban environment of "Slothsberg", a new highway ("Snailpace Highway") is to be constructed over the existing "Dillydally Highway". The following conditions apply to the...
-
Name eight or more ways to attract attention in opening a sales message.
-
Several costs incurred by Bay view Hotel and Restaurant are given in the following list. For each cost, indicate which of the following classifications best describe the cost. More than one...
-
Refer to information in QS 21-14. Compute the overhead volume variance for November and classify it as favorable or unfavorable. Data From QS 21-14 AirPro Corp. reports the following for November....
-
Hassellhouf Companys trial balance at December 31, 2017, is presented below. All 2017 transactions have been recorded except for the items described as unrecorded transactions. Unrecorded...
-
8. Two dice are rolled. Let A represent rolling a sum greater than 7. Let B represent rolling a sum that is a multiple of 3. Determine n(AB). A. 5 B. 8 C. 12 D. 15 Long Answer: Show all work for the...
-
Use the potential of the galvanic cell, Co(s) Co 2+ 'IIPb 2+ IPb(s), to determine Gf(Pb 2+ ), given that Gf(Co 2+ ) = 54.4 kJ mol 1 .
-
Which of the following reactions is (are) spontaneous at standard conditions? 3+ 2+ (a) Zn(s) + 2 Fe+ (aq) Zn+ (aq) + 2 Fe+ (aq) (b) Cu(s) + 2 H+ (aq) Cu+ (aq) + H(g) (c) 2 Br (aq) + I(s) Br() +2...
-
An advantage of a returns-based style analysis is that such analysis: A. is comparable across managers. B. is suitable for portfolios that contain illiquid securities. C. can effectively profile a...
-
Under certain conditions, the number of bacteria present in a colony is approximated by f(t) = A0e0.026t, where t is in minutes. If A = 2,600,000, find the number of bacteria present at 5 minutes, 10...
-
Do you agree we can predict someone reaction based on understanding his values`
-
4. Does the following graph G have a perfect matching? explain
-
1. What did Nadella bring in terms of his own background (professional and personal) that helped him to be successful in stepping into the CEO role? 2: What makes Nadella an "authentic leader"? What...
-
You have $500,000 saved for retirement. Your account earns 9% interest. How much will you be able to pull out each month, if you want to be able to take withdrawals for 15 years? $
-
Solve Problems 121 using ips units. AS 568A No. 320
-
For all of the following words, if you move the first letter to the end of the word, and then spell the result backwards, you will get the original word: banana dresser grammar potato revive uneven...
-
The 60-mm-diameter shaft is made of 6061-T6 aluminum having an allowable shear stress of Ï allow = 80 MPa. Determine the maximum allowable torque T. Also, find the corresponding angle of twist...
-
The A-36 steel shaft has a diameter of 50 mm and is subjected to the distributed and concentrated loadings shown. Determine the absolute maximum shear stress in the shaft and plot a graph of the...
-
The splined ends and gears attached to the A992 steel shaft are subjected to the torques shown. Determine the angle of twist of end B with respect to end A. The shaft has a diameter of 40 mm. 400 N-m...
-
Sam owns a courier business and on 1 November 2022 purchased a new van for $42,000 which has an effective life of 8 years. To help fund the purchase of the new van Sam traded his old one for $8,000....
-
How Close Revenues out to a temporary income summary account? How Close Assets out to a temporary income summary account effect on company's income?
-
Determine the pressure and force at the bottom of a 1m diameter open tank if it contains layers of 20 cm of oil, 60 cm of water and 10 cm of mercury. Given that the densities of oil, water and...

Study smarter with the SolutionInn App


