Which of the following reactions is (are) spontaneous at standard conditions? 3+ 2+ (a) Zn(s) + 2
Question:
Which of the following reactions is (are) spontaneous at standard conditions?
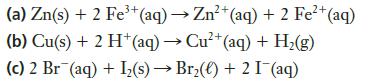
Transcribed Image Text:
3+ 2+ (a) Zn(s) + 2 Fe³+ (aq) → Zn²+ (aq) + 2 Fe²+ (aq) (b) Cu(s) + 2 H+ (aq) → Cu²+ (aq) + H₂(g) (c) 2 Br (aq) + I₂(s)→ Br₂() +2 I¯(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The reaction a Zns 2 Fe3aq Znaq 2 Feaq is not spontaneous at standard conditions ...View the full answer

Answered By

Gauri Hendre
I worked as EI educator for Eduphy India YT channel. I gave online tutorials to the students who were living in the villages and wanted to study much more and were preparing for NEET, TET. I gave tutions for topics in Biotechnology. I am currently working as a tutor on course hero for the biochemistry, microbiology, biology, cell biology, genetics subjects. I worked as a project intern in BAIF where did analysis on diseases mainly genetic disorders in the bovine. I worked as a trainee in serum institute of India and Vasantdada sugar institute. I am working as a writer on Quora partner program from 2019. I writing on the topics on social health issues including current COVID-19 pandemic, different concepts in science discipline. I learned foreign languages such as german and french upto A1 level. I attended different conferences in the science discipline and did trainings in cognitive skills and personality development skills from Lila Poonawalla foundation. I have been the member of Lila poonawalla foundation since 2017. Even I acquired the skills like Excel spreadsheet, MS Office, MS Powerpoint and Data entry.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Which of the following reactions are spontaneous in the direction written? See Table 18.2 for data. a. C(graphite) + 2H2(g) CH4(g) b. 2H2(g) + O2(g) 2H2O(l ) c. 4HCN(g) + 5O2(g) 2H2O(l) + 4CO2(g)...
-
A disproportionation reaction involves a substance that acts as both an oxidizing agent and a reducing agent, producing higher and lower oxidation states of the same element in the products. Which of...
-
From the values of H and S, predict which of the following reactions would be spontaneous at 25C: Reaction A: H 5 10.5 kJ/mol, DS 5 30 J/K ? mol; reaction B: H 5 1.8 kJ/mol, S 5 2113 J/K ? mol. If...
-
Use the ideas of Richardson's extrapolation and Romberg's method to evaluate the first derivative of f = ex at x = 0. To construct the first column of the Romberg's table, use the formula (exth -...
-
What could be included in an effective opening and closing of a claim or complaint letter?
-
The Department of Natural Resources is responsible for maintaining the states parks and forest lands, stocking the lakes and rivers with fish, and generally overseeing the protection of the...
-
Explain several implications of IFRS on financial reporting by health care organizations.
-
Sevilla Consulting offers environmental consulting services worldwide. The managers of branch offices are rewarded for superior performance with bonuses based on the economic value that the office...
-
At the Natural History Museum, tickets cost $10 for adults and $6 for kids under 12. What would be the total cost for 12 adult tickets and 18 kids tickets? What would be the total cost for a adult...
-
Consult a table of standard reduction potentials and determine which of the following reactions are spontaneous under standard electrochemical conditions. 2+ (a) Mn(s) + 2 H+ (aq) H(g) + Mn+ (aq)...
-
Suppose that you cannot find a table of standard reduction potentials. You remember that the standard reduction potential of Cu 2+ + 2 e Cu(s) is 0.337 V. Given that Gf(Cu 2+ ) = 65.49 kJ mol 1 and...
-
The following trial balance was prepared from the ledger accounts of Zumba Company: When the trial balance failed to balance, the accountant reviewed the records and discovered the following errors:...
-
There are three levels of analysis in OB, and as we move from the individual level to the national level to the global level, we add systematically to our understanding. Do you agree with this...
-
You want to be able to withdraw $20,000 each year for 30 years. Your account earns 7% interest. a) How much do you need in your account at the beginning? $ b) How much total money will you pull out...
-
(a1, a2,..., Consider the following recursive algorithm, which takes as input a sequence , an) of n numbers, where n is a power of two, i.e., n = 2k for some integer k > 0: Algorithm MYSTERY (a1,...
-
A tower tower that is 200 m tall is leaning to one side. From a certain point on the ground on that side, the angle of elevation to the top of the tower is 70. From a point 55 m closer to the tower,...
-
In 1999, the population of Richmond-Petersburg, Virginia, metropolitan area, was 961,400 and was increasing at a at roughly 9200 people per year. The average annual income in the area was $30,593 per...
-
Solve Problems 120 using ips units. AS 568A No. 160
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
The solid shaft of radius c is subjected to a torque T at its ends. Show that the maximum shear strain in the shaft is γ max = T c /JG. What is the shear strain on an element located at...
-
The propellers of a ship are connected to an A-36 steel shaft that is 60 m long and has an outer diameter of 340 mm and inner diameter of 260 mm. If the power output is 4.5 MW when the shaft rotates...
-
A motor delivers 500 hp to the shaft, which is tubular and has an outer diameter of 2 in. If it is rotating at 200 rad/s, determine its largest inner diameter to the nearest 1/8 in. if the allowable...
-
James Company is paid $6,000 in dividends from Mark Corp. on its equity investment. James lacks significant influence over Mark Corp. James Company should debit investment in trading securities debit...
-
Discuss the process of identifying which technologies are most important for an organization.
-
How would you respond to a question about why it is important to keep good financial records?

Study smarter with the SolutionInn App


