Identify what is incorrect in the Lewis structures shown for BBr 3 and SO 2 . (a)
Question:
Identify what is incorrect in the Lewis structures shown for BBr3 and SO2.
(a) 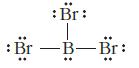
(b) 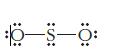
Transcribed Image Text:
: Br: : Br -B- Br:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a Br b Br Incorrect S Incorrect Br Br Br correct correct Br E...View the full answer

Answered By

Aqib Parvej
I am teaching since my graduation time so I have teaching experience of about 5 years and in these years I learn to teach in the best and interesting way .
4.80+
20+ Reviews
41+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Knowledge Management System Overview Objectives: How will you fill the knowledge gap by replacing existing knowledge? What methods will be used to increase accessibility and availability of...
-
Identify what is incorrect in the Lewis structures shown for O 3 and XeF 4 . (a) (b) |:000:
-
As you might imagine, the chart of accounts for a manufacturing firm would be different from that of a service firm. Not surprisingly, service firms differ so much that software now exists for almost...
-
What is capital rationing, what conditions lead to it, and how should it be dealt with?
-
Bioway, Inc., a manufacturer of medical supplies, uses aggregate planning to set labor and inventory levels for the year. While a variety of items are produced, a standard kit composed of basic...
-
In 2016, a hacker tricked a Lamps Plus employee into disclosing the tax information of approximately 1,300 other employees, resulting in the filing of a fraudulent income tax return of Lamps Plus...
-
Beagle Clothiers uses a weighted score for the evaluation and selection of its suppliers of trendy fashion garments. Each supplier is rated on a 10-point scale (10 = highest) for four different...
-
Convert the following Conceptual Model to Relational Model. STUDENT *student_id student_name student_address fills SEAT *seat_no seat position K takes COURSE *course_name *course_number has CLASS...
-
Which of the species listed has a Lewis structure with only one lone pair of electrons? F 2 , CO 3 2 , CH 4 , PH 3.
-
How does the existence of resonance structures for a molecule such as benzene explain its unusually unreactive chemical behavior?
-
Given that assets X and Y are perfectly correlated such that Y = 6 + .2X and the probability distribution for X is Probability X(%) .1........................30 .2........................20...
-
You have a 25% off coupon for a local store. You want to purchase an item that costs $105. a. What amount will you save by using the coupon? Include units.
-
A usb stick has a useful life that is exponential with a mean of 222 days. Determine the probability that any given unit will last between 38 days and 242 days.
-
Assume that (Ai , Bi) for I = 1, 2, . . . , n is a succession of free and indistinguishably dispersed 2-layered irregular factors where Ai and Bi are autonomous with minor disseminations Ai U[, ]...
-
How can Organizational Citizenship Behavior be used for organizational goal attainment?
-
mickey has a total of 955.there are a total of 133 bills consisting of 5 bills and 10 bills. How many of each type of bills does mickey have.
-
A company reportspretax accounting incomeof $10 million, but because of a single temporary difference,taxable incomeis $12 million. No temporary differences existed at the beginning of the year, and...
-
Why did management adopt the new plan even though it provides a smaller expected number of exposures than the original plan recommended by the original linear programming model?
-
Calculate the dipole moment of HF for the bonding MO in Equation (23.33). Use the method outlined in Section 23.8 to calculate the charge on each atom. The bond length in HF is 91.7 pm. The...
-
Evaluate the energy for the two MOs generated by combining two H1s AOs. Carry out the calculation for S 12 = 0.15, 0.30, and 0.45 to mimic the effect of decreasing the atomic separation in the...
-
Follow the procedure outlined in Section 23.2 to determine c u in Equation (23.17).
-
What are the mechanisms through which ethnic communities maintain cultural traditions and practices, and how do these processes contribute to the resilience and continuity of ethnic identities over...
-
Our Accounts Clerk prepared an acquisition analysis and calculated a gain on bargain purchase of $160,000 (being $112,000 cash paid less the subsidiary's equity acquired of $272,000) to be reported...
-
Explain the importance of analyzing financial performance reports.

Study smarter with the SolutionInn App


