Identify what is incorrect in the Lewis structures shown for O 3 and XeF 4 . (a)
Question:
Identify what is incorrect in the Lewis structures shown for O3 and XeF4.
(a) 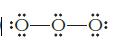
(b) 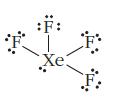
Transcribed Image Text:
|:0—0—0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a There are too many valence elect...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Knowledge Management System Overview Objectives: How will you fill the knowledge gap by replacing existing knowledge? What methods will be used to increase accessibility and availability of...
-
Identify what is incorrect in the Lewis structures shown for BBr 3 and SO 2 . (a) (b) : Br: : Br -B- Br:
-
The following data are for four independent process-costing departments. Inputs are added uniformly. Required: Compute the equivalent units of production for each of the preceding departments using...
-
In what sense is a reinvestment rate assumption embodied in the NPV, IRR, and MIRR methods? What is the assumed reinvestment rate of each method? Discuss.
-
What options are available for altering the capacity of (a) An elementary school (b) A prison (c) An airline?
-
On April 23, 2014, Calvin Loyer admitted his wife, Edeltrud Loyer, to a nursing home administered by Signature Healthcare. During the admissions process, Calvin signed an arbitration agreement...
-
Rufus and Rhonda are a married couple with 3 dependent children, all under 16 years of age. Rufus, 46, is an executive with Plowshare Corporation. Rhonda, 39, is a self-employed attorney. Rufus...
-
Breast cancer patients in a London, England, hospital were being treated for spinal metastases. They were followed over a five-year period, with their ambulatory state being recorded before treatment...
-
Chemical species are said to be isoelectronic if they have the same Lewis structure (regardless of charge). Consider these ions and write a Lewis structure for a neutral molecule that is...
-
Which of the species listed has a Lewis structure with only one lone pair of electrons? F 2 , CO 3 2 , CH 4 , PH 3.
-
Define the following terms: a. Asymmetric information b. Adverse selection c. Moral hazard d. Principal-agent problem
-
A doctor wants to test the relationship between the daily consumption of cigarettes (variable X) and the number of sleep hours (variable Y). The following data shows the sums of the study for a...
-
Consider the following function. f(x)=9x^(4)-7x^(2) Determine whether the function is even, odd, or neither. f(x) is even. f(x) is odd. f(x) is neither even nor odd. Find the zeros of the function.
-
What are the disadvantages and advantages of using demographic segmentation? Can demographic segmentation and psychographics be used together to determine target markets? Give a specific example a...
-
Lime Spring Select your answer (13 out of 20) Where does the polynomial y=x^(2)-3x+2 cross the x-axis?
-
Consider the Library Java Project posted on Moodle. Download the file and run the project using Eclipse. You can download Eclipse for Windows using this link:...
-
Refer to the situation described in BE 16-4. Suppose the unearned portion of the rent collected was $40 million at the end of 2012. Taxable income is $200 million. Prepare the appropriate journal...
-
In Exercises find dy/dx by implicit differentiation. xy - y = x
-
A HartreeFock calculation using the minimal basis set of the 1s, 2s, 2p x , 2p y , and 2p z AOs on each of N and O generated the energy eigenvalues and AO coefficients listed in the following table....
-
Calculate the value for the coefficients of the AOs in Example Problem 23.4 for S 12 = 0.45. How are they different from the values calculated in that problem for S 12 = 0.3? Can you offer an...
-
Using the method of Mulliken, calculate the probabilities of finding an electron involved in the chemical bond on the H and F atoms for the bonding and anti-bonding MOs for Problem P23.9.
-
To save on future home insurance costs, you need a new roof and a burglar alarm system that will cost $10,000 five years from now. You can earn 3 percent on your savings. How much should you deposit...
-
Jamie and Ross decided to conduct a checkup on their homeowners insurance policy. They noticed that they had omitted covering Jamie Lees diamond wedding band set from their policy. What if it got...
-
Medicare and Medicaid are federal programs that cover a large portion of the US population with healthcare. The two have Similar Objectives in the area of providing care, but there are significant...

Study smarter with the SolutionInn App


