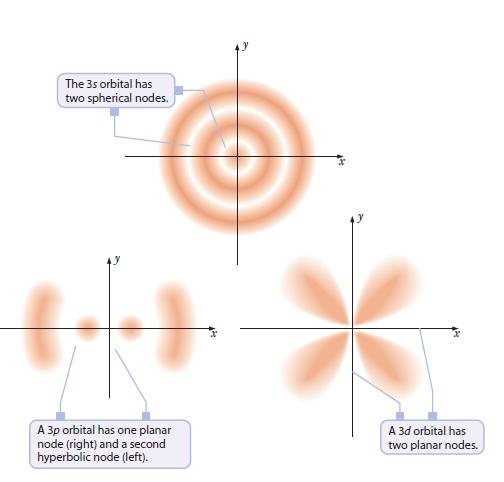
Referring to Figure 6.15, draw a 4p orbital, showing all of its nodes. Figure 6.15 The 3s
Question:
Referring to Figure 6.15, draw a 4p orbital, showing all of its nodes.
Figure 6.15
Transcribed Image Text:
The 3s orbital has two spherical nodes. + A 3p orbital has one planar node (right) and a second hyperbolic node (left). A 3d orbital has two planar nodes.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
The illustration of t...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Determine the force in each member of the space truss in E9.3.27 if the magnitudes of F and F are 8 kip and 4 kip, respectively. State whether each member is in tension or compression. 2 ft F2 2 ft...
-
A certain shoe conies in 5 different styles with each style available in 4 distinct colors. If the store wishes to display pairs of these shoes showing all of its various styles and colors, how many...
-
The first payroll in October covered the two workweeks that ended on September 26 and October 3. This payroll transaction has been entered for you in the payroll register, the employees' earnings...
-
Discuss the differences between @classmethod , @staticmethod , and instance methods in Python.
-
The Dinkle and Frizell Dental Clinic provides both preventive and orthodontic dental services. The two owners, Reese Dinkle and Anita Frizell, operate the clinic as two separate investment centers:...
-
Soundgarden Company sold 200 copymaking machines in 2008 for $4,000 apiece, together with a one-year warranty. Maintenance on each machine during the warranty period averages $330. Instructions (a)...
-
A project has been selected for implementation. The net cash flow (NCF) profile associated with the project is shown below. MARR is 10 percent/year. a. What is the annual worth of this investment? b....
-
At week 24 of a project to shoot a television commercial, the project manager is worried about her budget since costs have risen to $7,500. Is there a cost overage? If so, how much is it? Is the...
-
(a) Consider the recurrence below defined for n 0. 3 T(n): if n = 0 7T (n - 1) +12 if n > 0 Give a closed-form solution to the recurrence. You only have to give the solution. You do not need to show...
-
Define the term nodal plane (or node).
-
Tungsten alloys are often used for parts that must withstand high temperatures. Use the periodic table to determine the electron configuration of tungsten (W). Strategy Begin by finding tungsten in...
-
True or false The APV method a. Starts with a base-case value for the project. b. Calculates the base-case value by discounting project cash flows, forecasted assuming all-equity financing, at the...
-
Explain how each of the following may contribute to the effectiveness of monetary policy: establishing news conferences following some FOMC meetings; announcing that the FOMC has an inflation goal of...
-
If I receive a 5% increase in wages and the inflation rate is 5%, am I better off, worse off, or the same? Explain
-
Investment A returns 2400 in t = 6. Investment B returns 2800 in t = 8. At what interest rate do both investments have the same present value in t = 0 (Give your answer as a decimal, i.e. answer 0.06...
-
Ricky quits his job earning $150,000/year and converts a house that he owns into a museum for his large collection of antique clocks. Before this, Ricky had been renting the house out for...
-
How are the structure and function of global societies and global cultures similar and different than global economies and global politics?
-
What impact would you expect each of the following events to have on business cycles? Label each as a demand-side or supply-side shock. a. Defense production increases due to the imminent threat of...
-
A 2500-lbm car moving at 15 mi/h is accelerated at a constant rate of 15 ft/s 2 up to a speed of 50 mi/h. Calculate force and total time required?
-
Identify the number of steps (patterns) for the mechanisms in Problems. For example, the patterns for the first two are: a: ±H ± LG Nuc attack This mechanism exhibits a proton transfer...
-
Consider the structure of the compound below. (a) Identify each position where an S N 2 reaction is likely to occur. (b) Identify each position where an S N 1 reaction is likely to occur. Cl CI TsO....
-
Does each of the following solvents favor an SN2 reaction or an SN1 reaction? a) b) c) d) (e) MeOH (f ) CH 3 CN (g) HMPA (h) NH 3 %24
-
Multiple Choice Question Which group of costs is the most accurate example of variable cost? Multiple choice question. Payments for materials, fuel, and transportation services Payments for power,...
-
A firefighter, whose mass (including clothing and equipment) is m= 92 kg, hears the alarm and slides down the pole with a constant downward acceleration of magnitude a=3.04 m/s 2 What is the...
-
According to the information presented below, can you calculate the Debt-to-Equity Ratio for Alternatives, INC? *Round your answer to the nearest two decimals, if needed. If the answer is a...

Study smarter with the SolutionInn App


