The following rate constants were obtained in an experiment in which the decomposition of gaseous N 2
Question:
The following rate constants were obtained in an experiment in which the decomposition of gaseous N2O5 was studied as a function of temperature. The products were NO2 and NO3.
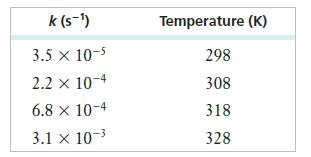
Determine Ea for this reaction in kJ/mol.
Transcribed Image Text:
k (S-¹) 3.5 x 10-5 2.2 x 10-4 6.8 x 10-4 3.1 x 10-³ Temperature (K) 298 308 318 328
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
E ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
As the air temperature drops, river water becomes supercooled and ice crystals form. Such ice can significantly affect the hydraulics of a river. The article "Laboratory Study of Anchor Ice Growth"...
-
The reaction C 2 H 5 I + OH - C 2 H 5 OH + I - was studied in an ethanol (C 2 H 5 OH) solution, and the following rate constants were obtained: 15.83 C, k = 5.03 x 10 -5 ; 32.02 C, 3.68 x 10 -4 ;...
-
For the reaction I (aq) + OCl (aq) OI (aq) + Cl (aq) occurring in aqueous solution, the following mechanism has been proposed: a. Derive the rate law expression for this reaction based on this...
-
A house girl added 20g of sodium chloride (NaCl) to 80g of water (atomic masses are Na=23amu, Cl=35.5amu). Calculate a)Percent(w/w) of NaCl b)Mole fraction of NaCl
-
On January 1, the company had 150,000 common shares outstanding. During the year, the following events occurred: March 1: 2-for-1 stock split June 1: Issued 45,000 additional shares September 1: 20%...
-
Near the end of 2011, the management of Simid Sports Co., a merchandising company, prepared the following estimated balance sheet for December 31, 2011. To prepare a master budget for January,...
-
How does make determine whether a target needs to be rebuilt?
-
Suppose that every driver faces a 1% probability of an automobile accident every year. An accident will, on average, cost each driver $10,000. Suppose there are two types of individuals: those with...
-
Country A is endowed with 18 units of good x and 1 unit of good y, while country B is endowed with 2 units of good x and 7 units of good y. Both countries consume x and y in fixed and equal...
-
The table below presents measured rate constants for the reaction of NO with ozone at three temperatures. From these data, determine the activation energy of the reaction in kJ/mol. (Assume the...
-
Use the kinetic-molecular theory to explain why an increase in temperature increases reaction rate.
-
A circle of radius 3 is inscribed in a square. What is the product of the perimeter and area of the square?
-
How many times is the text "Let us C" printed if the code snippet given below is run? int i = 0; do { cout < < "Let us C" < < endl; i++; if (i 2 == 0) { i = 11; } } while (i
-
4) A proposed new investment has projected sales of $515,000. Variable costs are 37 percent of sales, and fixed costs are $133,000; depreciation is $52,000. Prepare a pro forma income statement...
-
You have been asked to build a web application for your local soccer club which will record and display fixtures for matches. A match fixture should contain information about the competition (such as...
-
A pendulum makes 42 cycles in 50.0 seconds. What is its period? What is its frequency?
-
Can you elucidate the role of strategic foresight and horizon scanning in long-term planning processes, including the identification of emerging trends, disruptive technologies, and market...
-
Hearts Inc. acquired the following assets in January 2012. Equipment, estimated service life, 5 years; no salvage value ......$650,000 Building, estimated service life, 40 years; salvage value ,...
-
4. Jobe dy -Y 2 et by
-
Oxygen sensing is important in biological studies of many systems. The variation in oxygen content of sapwood trees was measured by del Hierro and coworkers [ J. Experimental Biology 53 (2002): 559]...
-
The pyrene/coumarin FRET pair (r 0 = 39 ) is used to study the fluctuations in enzyme structure during the course of a reaction. Computational studies suggest that the pair will be separated by 35 ...
-
In a FRET experiment designed to monitor conformational changes in T4 lysozyme, the fluorescence intensity fluctuates between 5000 and 10,000 counts per second. Assuming that 7500 counts represent a...
-
What is a substantial response to this question? Is the good faith exception abused by law enforcement?
-
Explain when and why a defendant may move for judgment as a matter of law in the United States.?
-
in law enforcement , what can be done to ensure identifications are accurate? Please explain

Study smarter with the SolutionInn App


