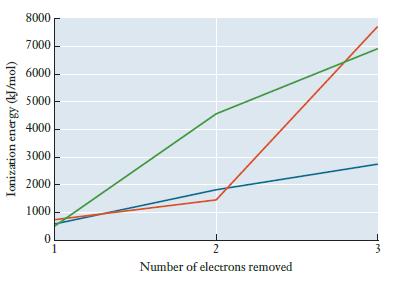
The graph below shows the first three ionization energies for sodium, magnesium, and aluminum. Without consulting a
Question:
The graph below shows the first three ionization energies for sodium, magnesium, and aluminum. Without consulting a list of values, determine which line in the graph corresponds to each element.
Transcribed Image Text:
Ionization energy (kJ/mol) 8000 7000 6000 5000 4000 3000 2000 1000 0 2 Number of electrons removed 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Green ...View the full answer

Answered By

Muhammad Haroon
More than 3 years experience in teaching undergraduate and graduate level courses which includes Object Oriented Programming, Data Structures, Algorithms, Database Systems, Theory of Automata, Theory of Computation, Database Administration, Web Technologies etc.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
A particular element has the following values for its first four ionization energies: 900, 1760, 14,850, and 21,000 kJ/mol. Without consulting a list of ionization energy values, determine what group...
-
The graph below shows a production possibilities curve for 2014 and two potential production possibilities curves for 2015, denoted 2015A and 2015B. a. Which of the labeled points corresponds to...
-
The graph below shows the hours studied and the test grades on a biology test for 7 students. The red line on the graph can be used to approximate the test grade the average student receives for the...
-
describe Jarir bookstore by providing the followings: - A brief introduction regarding the history of this brand and its main activity? - Describe how and why the brand's activity has evolved through...
-
Stiever Company estimates that variable costs will be 60% of sales, and fixed costs will total $800,000. The selling price of the product is $4. Instructions (a) Prepare a CVP graph, assuming maximum...
-
Felicia Rashad Corporation has pretax financial income (or loss) equal to taxable income (or loss) from 2000 through 2008 as follows. Pretax financial income (loss) and taxable income (loss) were the...
-
You need to understand the approach described in question 3 in More Genetic TIPS before answering this question. A muscle-specific gene was cloned and then subjected to promoter bashing. As shown...
-
Charter Corp. has issued 2,500 debentures with a total principal value of $2,500,000. The bonds have a coupon interest rate of 7%. a. What dollar amount of interest per bond can an investor expect to...
-
Solving via linear equation No unread replies.No replies. A small country exports soybeans and flowers. Soybeans require 8 workers per acre, flowers require 12 workers per acre, and 100,000 workers...
-
Which graph correctly depicts the first ionization energy of three elements in groups 14 (dashed line) and 17 (solid line)? Explain the reasoning you used to make your choice. 1st I.E. Si Br Ge 1st...
-
Why is there no element to the immediate right of magnesium in the periodic table?
-
A presidential candidate is concerned that Americans create too much pollution by driving excessively. She proposes issuing a fixed number of tradable driving permits each month. Each permit would be...
-
Here, an object of mass m is at the bottom of a hill (A) as shown in the figure. The object is making attempts to cross over both the points B and D in order to go to E against gravity. Consider that...
-
Techfin Consulting, a profitable company, has yet to pay dividends. However, its board has indicated to initiate a $0.25 dividend in 4 years and increase by 30% each year for 2 years before reaching...
-
1 FSMA 300 Quiz 1-Final- 3ed- Protected View - Saved to this PC Layout References Mailings 42. Revenue bonds are A: ABCDE B: C: D: E: A: B: C: D: E: Review ABCDE View 43. Jason is in the 28% Federal...
-
The annualized Indian rupee (INR) interest rate is 10% for six months, while the annualized USD interest rate is 3% for six months. Khaledi Co., a U.S. firm, entered into a currency swap with a swap...
-
There is a compressed spring between two laboratory carts of masses m = 175 g and m = 222 g. Initially, the carts are held at rest on a horizontal track as shown in figure A. The carts are released,...
-
How will inflation impact the currency of India and its trade opportunities?
-
Solve each equation or inequality. |6x8-4 = 0
-
The molar volume of H 2 O(l) decreases with increasing temperature near 4C. Can you explain this behavior using a molecular level model?
-
For each compound below, identify any polar covalent bonds, and indicate the direction of the dipole moment using the symbols + and -: a. HBr b. HCl c. H 2 O d. CH 4 O
-
Why was the following qualification made in Section 3.7? Note that Equation (3.47) is only applicable to a process in which there is no change in the phase of the system, such as vaporization or...
-
Java and Python are common programming languages used by organizations. At a party, you meet a lady who works at Exxon. She tells you that she writes software using Java. What is her job role?
-
Class A IP addresses has a range of : 1-126 128-191 240-255 224-239
-
Lakson investment Company holds an option on HBL's stock. The contract multiplier is 500 shares with a exercise price of PKR70 per share. The option's expiration date is from 1st January 2024 to 30th...

Study smarter with the SolutionInn App


