The picture shown depicts the species present at the start of a combustion reaction between methane, CH
Question:
The picture shown depicts the species present at the start of a combustion reaction between methane, CH4, and oxygen, O2.
(a) What is the limiting reactant?
(b) Draw the resulting state after this set of reactants has reacted as far as possible.
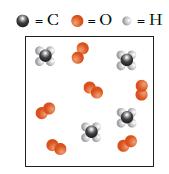
Transcribed Image Text:
= C = 0 = H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
To determine the limiting reactant we need to consider the stoichiometry of the combustion reaction ...View the full answer

Answered By

Amar Kumar Behera
I am an expert in science and technology. I provide dedicated guidance and help in understanding key concepts in various fields such as mechanical engineering, industrial engineering, electronics, computer science, physics and maths. I will help you clarify your doubts and explain ideas and concepts that are otherwise difficult to follow. I also provide proof reading services. I hold a number of degrees in engineering from top 10 universities of the US and Europe.
My experience spans 20 years in academia and industry. I have worked for top blue chip companies.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
The picture shown depicts the species present at the start of a combustion reaction between methane, CH 4 , and oxygen, O 2 . (a) Draw the resulting state after this set of reactants has reacted as...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Methane combustion on La2O3-based catalysts has been studied by Toops et al. [46]. With a 4% Sr-promoted La2O3 catalyst (2:5m2g-1) operating between 773 and 973 K, 0:5 -5 Torr CH4 and 3 _ 23 Torr O2...
-
1/ You just started working and you planned to save $5,000 every year in your retirement account. How much money will you have in your retirement account once you retire in 40 years? Your retirement...
-
Leonard Industries wishes to prepare a pro forma balance sheet for December 31, 2013. The firm expects 2013 sales to total $3,000,000. The following information has been gathered. (1) A minimum cash...
-
The traditional income statement for Pace Company shows sales $900,000, cost of goods sold $600,000, and operating expenses $200,000. Assuming all costs and expenses are 70% variable and 30% fixed,...
-
A U.S. Treasury bond pays a 7% coupon on January 7 and July 7. How much interest accrues per \($100\) of principal to the bond holder between July 7, 2013, and August 9, 2013? How would your answer...
-
Included in the December 31, 2006 Jacobi Company balance sheet was the following stockholders equity section: The company engaged in the following stock transactions during 2007: Jan. 2 Paid the...
-
8. The electronic health record shows an order for 120 mL of 25 % formula to be administered in the nasogastric feeding tube every 2 hours between 0700 and 1900 each day. The 30 mL can of...
-
New chemistry students sometimes try to use the dilution equation, M 1 V 1 = M 2 V 2 to solve solution stoichiometry problems. (a) What concept is being overlooked by a student who takes this...
-
The particulate scale drawing shown depicts the products of a reaction between N 2 and O 2 molecules. (a) Draw a similar representation for the reactants that must have been present before the...
-
1. Why are the views of the German managers and associates so different? What are the underlying cultural values that may be causing the differences? Why are views so different can be discussed based...
-
1. Find the slope-intercept form of the line which passes through the given points (-2,4) and (4,-2). 2. Find the line perpendicular to the given line y = -3x + 7 which passes through the point...
-
Complete all three questions. Show as much work as possible. 1. Solve. sin0-sin0 = 0 2. Determine whether the function f(x)=x-1 is one-to-one. If it is, find it's inverse function. 3. Solve....
-
In 1905, Betsy was a 25-year-old single woman living in New York. Her father gave Betsy a small farm and farmhouse. Betsy married William in the year 1910. Betsy would like to sell the farm to...
-
Provide an analysis of In the Aftermath of R v Pham: A Comment on Certainty of Removal and Mitigation of Sentences" by Sasha Baglay. Be sure to answer each of the following questions: What is Sashas...
-
10 If [0 f '10 60 9 f(x) dx = 35 and g(x) dx = 18, find 10 6. [2f(x) + 3g(x)] dx.
-
Describe a work report that you are required (or have been required) to complete on a regular basis. How would you present the information related to this report in a visual manner? Would a visual...
-
a. Determine the domain and range of the following functions.b. Graph each function using a graphing utility. Be sure to experiment with the window and orientation to give the best perspective of the...
-
Draw a plausible mechanism for each of the following transformations: (a) (b) (c) (d) Meo, OMe [H,SO4] excess MeOH -H20
-
Draw a plausible mechanism for each of the following reactions: (a) (b) [H,SO4] -H20 [H,SO4] -H20
-
Draw a plausible mechanism for each of the following reactions: (a) (b) HO HO- [H2SO4] -H20
-
A 2200 kg car is moving at a speed of 31.0 m/s. Determine the stopping distance (in meters) if the brakes supply a force of 3500 newtons.
-
Hunter the "wonder dog" is going to dive into a pool of water from a 10.0 m cliff. Hunter weighs 180.0 N. How much kinetic energy will Hunter have when he reaches the water with his dive?
-
The transmission line is terminated with 50 load resistor. Power with is delivered into this load is -30 dBm. Calculate the magnitude of voltage drop across this load resistor

Study smarter with the SolutionInn App


