Using only the data given below, determine G for the following reaction: NO(g) + O(g) NO(g)
Question:
Using only the data given below, determine ΔG° for the following reaction:
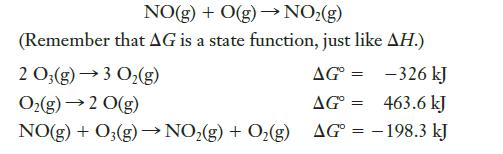
Transcribed Image Text:
NO(g) + O(g) → NO₂(g) (Remember that AG is a state function, just like AH.) 2 O3(g) →3 O₂(g) AG= -326 kJ O₂(g) → 2 0(g) AG = 463.6 kJ NO(g) + O₂(g) →→NO₂(g) + O₂(g) AG = -198.3 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To determine G for the reaction NOg Og NO2gwe can use the following Hesss Law cycl...View the full answer

Answered By

Aun Ali
I am an Associate Member of Cost and Management Accountants of Pakistan with vast experience in the field of accounting and finance, including more than 17 years of teaching experience at university level. I have been teaching at both undergraduate and post graduate levels. My area of specialization is cost and management accounting but I have taught various subjects related to accounting and finance.
5.00+
13+ Reviews
32+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Using the data given below that relate to the formation of Schottky defects in some oxide ceramic (having the chemical formula MO), determine the following: (a) The energy for defect formation (in...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Using only the data given in the text, what is the maximum number of telephones that the existing U.S. system can support without changing the numbering plan or adding additional equipment? Could...
-
1. A drying process is used to remove 3000 lbm/h of water (vapor) at steady-state from processed paper. The dryer first heats atmospheric air (state 1) at 80F, 1 atm and 60% relative humidity to 250F...
-
Judge Daniel H. Wells is currently deliberating over a suit filed by three stockholders against Transcontinental Corporation. The stockholders allege that Transcontinentals year-end balance sheet was...
-
1. If a government runs a deficit, it will ____________its outstanding debt. 2. Proponents of Ricardian equivalence are primarily concerned about deficits crowding out the stock of capital....
-
In the organizational chart for the consumer-packaged goods firm in Figure 22 5, where do product line, functional, and geographical groupings occur? Figure 22-5 Chief Marketing Officer or Vice...
-
1. Create a detail report that will display all SCR courses in alphabetical order, with the course name and the instructor name in a group header; the Social Security number, name, and telephone...
-
In some ways, a qualitative portfolio manager could never really be an index portfolio manager, whereas a quantitative portfolio manager could be. Explain why.
-
The graph below shows G as a function of temperature for the synthesis of ammonia from nitrogen and hydrogen. (a) Use the graph to estimate S for the ammonia synthesis reaction. (b) Given that the...
-
The reaction shown below is involved in the refining of iron. (The table that follows provides all of the thermodynamic data you should need for this problem.) (a) Find H for the reaction. (b) S for...
-
Consider the following problem. Maximize Z = 2x1 + 4x2 + 3x3, subject to and x1 0, x2 0, x3 0. You are given the information that x 1 > 0, x 2 = 0, and x 3 = 0 in the optimal solution. (a)...
-
A systems owner reviews permissions granted to a targeted team of employees and determines that some of the employees' accesses are outside their scope of work. After revoking the certificates, what...
-
Identify different methods used in needs assessment and identify the advantages and disadvantages of each method.
-
Liam, a sole proprietor, contributed accounts receivable (basis $0 (zero), FMV $20,000), inventory (basis $25,000, FMV $50,000), and land (investment property to Liam, basis $12,000, FMV $9,000) to...
-
Work legally and ethically lactivity To provide you with an opportunity to use effective problem-solving techniques when exposed to competing value systems. What are the eight steps for an effective...
-
Walker & Daniels is an accounting firm which does a sizeable business in individual tax preparation and filing. The firm charges $250 for its tax services and maintains a 60% contribution margin....
-
Most decisions made by management impact the ratios analysts use to evaluate performance. Indicate (by letter) whether each of the actions listed below will immediately increase (I), decrease (D), or...
-
Prove that if Σ an is absolutely convergent, then a. an
-
Four bases (A, C, T, and G) appear in DNA. Assume that the appearance of each base in a DNA sequence is random. a. What is the probability of observing the sequence AAGACATGCA? b. What is the...
-
The natural abundance of 13 C is roughly 1%, and the abundance of deuterium ( 2 H or D) is 0.015%. Determine the probability of finding the following in a mole of acetylene: a. H- 13 C- 13 C-H b. D-...
-
In the neck of the flask depicted in the text, five red balls rest on five blue balls. Suppose the balls are tipped back into the flask, shaken, and the flask is re-inverted. Whats the probability...
-
a. What is the smallest unit of measurement on the ruler above? (e.g., millimeter, centimeter) b. How accurate do you think you can be when measuring an object with this ruler? Explain your answer....
-
6. [6 marks] Two forces act horizontally on a mass. F is 200 N [S25E]. F is 130 N [E15N]. a) Find the resultant force FR = F + F. b) Find the equilibrant force FE. c) Draw a vector diagram showing F,...
-
5. Two seekers, both intent on capturing the snitch fail to notice each other as they are flying through the air. The Gryffindor seeker (mass = 60.5 kg) is flying at a constant 15 m/s [35 S of W]...

Study smarter with the SolutionInn App


