What type of hybridization would be expected for the central atom X in each of the following
Question:
What type of hybridization would be expected for the central atom X in each of the following Lewis structures?
(a) 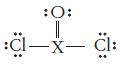
(b) ![]()
(c) 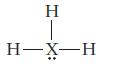
Transcribed Image Text:
:O: :Cl—x— Cl: -X-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a sp C1 ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Solve the right triangle. Round decimal answers to the nearest tenth. A 11 B 5 C
-
What hybrid orbitals would be expected for the central atom in each of the following molecules or ions? a. SeCl2 b. NO2 c. CO2 d. COF2
-
What hybrid orbitals would be expected for the central atom in each of the following molecules or ions? a. GeCl4 b. PBr3 c. BeF2 d. SO2
-
Consider the no-trade input/output situation presented in the following table for countries X and Y. Assuming that free trade is allowed, develop a scenario that will benefit the citizens of...
-
(1) Draw NPV profiles for Franchises L and S. At what discount rate do the profiles cross? (2) Look at your NPV profile graph without referring to the actual NPVs and IRRs. Which franchise or...
-
Tara McCoy is the office administrator for the Department of Management at State University. The faculty uses a lot of printer paper and Tara is constantly reordering and frequently runs out. She...
-
Mrs. Clarks Foods was an Iowa company engaged in the business of distributing juice beverages. International Suntrade and Miller & Smith Foods were Canadian companies that acted as brokers...
-
Dynabase Tool has forecast its total funds requirements for the coming year as shown in the following table. a. Divide the firm's monthly funds requirement into (1) A permanent component and (2) A...
-
Define the relational model. What is a relational database management system (DBMS)?
-
The number of hybrid orbitals formed must always be equal to the number of atomic orbitals combined. Suggest a reason why this must be true.
-
What observation about molecules compels us to consider the hybridization of atomic orbitals?
-
y 5 = 1,024, y = A. 5 B. 4 C. 3 D. 1/5 E. 1/4
-
A 50 year old pine tree is 40m tall. If pine trees can be expected to grow at an annual rate of approximately 0.5m per year, how tall was this tree when it was planted?
-
Goran asks his Accountant to falsify some figures in his tax return to ensure he pays less tax. He tells the accountant that he will pay him an extra $200 if he does this. Is this a valid contract?...
-
If an 11 per cent increase in the price of Kellogg's cereal causes a 12 per cent reduction in the number of boxes of cerealdemanded, what is the price elasticity of demand for Kellogg's cereal?...
-
When calculating cost per QALY, how are differences in patient income or wealth included in the analysis? The calculation of QALY?
-
The table gives mass and speed data for the two objects in the figure. Mass Speed Object A Object B 2.20 kg 6.62 kg 6.62 m/s 2.20 m/s VB Concepts: (i) Is it possible for two objects to have different...
-
First Bank has some question as to the tax-free nature of $5 million of its municipal bond portfolio. This amount is excluded from First Bank's taxable income of $55 million. Management has...
-
Show that every group G with identity e and such that x * x = e for all x G is abelian.
-
Using Figures 23.9 and 23.10, explain why Î Ï 2 g < 0 and ÎÏ 2 u > 0 outside of the bonding region of H + 2 . Figure 23.9 Figure 23.10 Bonding - Antibonding . , Bonding...
-
Using Figures 23.9 and 23.10, explain why 2 g < 0 and 2 u > 0 outside of the bonding region of H + 2 . Figure 23.9 Figure 23.10 Bonding Antibonding H.
-
Consider the molecular electrostatic potential map for the BeH 2 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
A nuclear power plant can produce large amounts of power by harnessing the heat produced by nuclear fission. That heat boils water, and the steam spins a turbine connected to a generator which...
-
Balance the following chemical reactions by placing the correct coefficient in front of the compounds. Be sure to show your work. Label the type of each chemical reaction. (7 points each) (provided...
-
It seems like we are surrounded by new innovations every day. However, if you think about it, you can probably list several industries, organizations, processes, etc. that are far behind the...

Study smarter with the SolutionInn App


