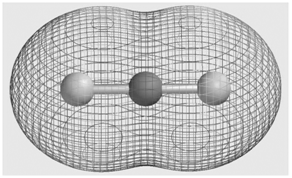
Consider the molecular electrostatic potential map for the BeH 2 molecule shown here. Is the hydrogen atom
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
It is an electron acceptor becaus...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the molecular electrostatic potential map for the BH 3 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the molecular electrostatic potential map for the NH 3 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the molecular electrostatic potential map for the LiH molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
The stockholders' equity accounts of Whispering Company have the following balances on December 31, 2025. Common stock, $10 par, 304,000 shares issued and outstanding $3,040,000 Paid-in capital in...
-
In Exercises 19 and 20, use a graphing utility to graph f, g, and y = x in the same viewing window to verify geometrically that g is the inverse function of f. (Be sure to restrict the domain of f...
-
The bank columns in the cash book for November 2004 and the bank statement for that month for S Shaw are: (a) Write up the cash book up to date and state the new balance as on 30 November 2004. (b)...
-
Why might a plaintiff wish to serve a demand letter on the defendant before filing a lawsuit?
-
The Public Safety office at Millennium College maintains a list of parking tickets issued to vehicles parked illegally on the campus. Table 4-6 shows a portion of this list for the fall semester....
-
On 20 April 2023, on the evening of the book launch party,Karolina snuck into Gigi's Fitzroy home.The main front gate was locked, and guests received an entry pin with their invitation to open the...
-
Ivanhoe Rook Candies received a bank statement for the month of August 2014, which showed a balance per bank of $6,964. The company's cash account in the general ledger showed a balance of $6,248 at...
-
Using Figures 23.9 and 23.10, explain why 2 g < 0 and 2 u > 0 outside of the bonding region of H + 2 . Figure 23.9 Figure 23.10 Bonding Antibonding H.
-
Why are MOs on hetero-nuclear diatomics not labeled with g and u subscripts?
-
The members of a population are numbered 1-50. Use Table I in Appendix A to obtain an SRS of size 6 from the population. Start at the two-digit number in line number 10 and column numbers 1011, read...
-
Name any four interactions in the tertiary structure of proteins.
-
Research legislation that has occurred within the last 5 years at the Texas or federal level as a result of nurse advocacy. Describe the legislation and what was accomplished. What additional steps...
-
PLEASE HELP ME MY CANCEL BUTTON AND TRY BUTTON DO THE SAME THING I WANT MY CANCEL BUTTON TO CLOSE MY PROGRAM/FRAME import java.awt. * ; import javax.swing. * ; import java.awt.event. * ; import...
-
BrockChilton is a small-medium auditing firm that specialises in auditing retirement villages. BrockChilton is currently auditing the accounts of Ventnor Retirement Village, a place where Jenny...
-
Capital assets are important in all businesses because they represent significant investments. How are capital assets accounted for in government and not-for-profit organizations? Provide examples....
-
Your assistant has just completed a rough draft of Form 941, shown on pages 3-72 and 3-73, for the quarter ending March 31, 20--. As the supervisor and authorized signer, you are auditing the form...
-
Havel says the grocer doesnt believe what is on the sign and indeed, he says the grocers customers will barely notice it. But Havel maintains that the sign serves a specific function. How would you...
-
Which of the following molecules may show a pure rotational microwave absorption spectrum: (a) H 2 , (b) HCl, (c) CH 4 , (d) CH 3 Cl, (e) CH 2 Cl 2 ?
-
The rotational Raman spectrum of 35 Cl 2 (m( 35 Cl) = 34.9688 u) shows a series of Stokes lines separated by 0.9752 cm 1 and a similar series of antiStokes lines. Calculate the bond length of the...
-
The wavenumber of the incident radiation in a Raman spectrometer is 20 487 cm 1 . What is the wavenumber of the scattered Stokes radiation for the J = 2 0 transition of 14 N 2 ?
-
Three not-for-profit organizations operate separately in Sunland City. The most established entity, Sunland Support, has been around for over 100 years and generates by far the largest amount of...
-
E. Chrissy's Vases is a boutique selling artisan vases. The boutique purchases several styles of vases from one source. The cost of the vases to the boutique is $10 each. All other costs for...
-
Hampton Muffins sells its muffins to restaurants and coffee houses for an average selling price of $26 per case. The following information relates to the budget for Hampton Muffins for this year (all...

Study smarter with the SolutionInn App


