Which of the following is more likely to precipitate sulfate ions? PbSO4(s) Pb+ (aq) + SO4 (aq)
Question:
Which of the following is more likely to precipitate sulfate ions?
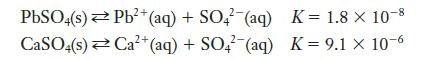
Transcribed Image Text:
PbSO4(s) Pb²+ (aq) + SO4 (aq) 2+ CaSO4(s) Ca²+ (aq) + SO4 (aq) K = 1.8 X 10-8 K = 9.1 x 10-6
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Lead is m...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
In Example Problem 12.2, we saw that hydroxide ions precipitate with calcium. Magnesium ions show similar behavior. The two pertinent equilibria are: Which ion is more likely to precipitate hydroxide...
-
Which of the following is more likely to precipitate the hydroxide ion? Cu(OH) (s) Ca(OH)(s) 2+ Cu+ (aq) + 2 OH(aq) Ca+ (aq) + 2 OH- (aq) K 1.6 X 10-19 K = 7.9 x 10-6
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
paraphrase the following two passages using narrative in-text citations: 1.Those who teach ethics don't need to look far for lessons. Every day there's fresh scandal: Google is in hot water for how...
-
Is Samsungs product development process customer centered? Team based? Systematic? In the world of consumer electronics, copycat brands are a dime a dozen. These are the brands consumers turn to if...
-
Matt Mouw is in charge of providing computing and other information technology services to his firm. Matts firm has four distinct product lines, each operated as a stand-alone business. Profit is the...
-
Nationwide Insurance developed a program to recruit new insurance agents by offering planning, training, and start-up financing to build self-sustaining agencies. These new agents would be...
-
Mike Dreskin manages a large Los Angeles movie theater complex called Cinema I, II, III, and IV. Each of the four auditoriums plays a different film; the schedule is set so that starting times are...
-
The KLM Partnership, which uses the accrual method of accounting, is owned equally by Karen ( cash method taxpayer ) , and LM corporation ( accrual method taxpayer ) . Karen is a real estate...
-
Write equilibrium expressions for each of the following heterogeneous equilibria. (a) CaCO 3 (s) Ca 2+ (aq) + CO 3 2 (aq) (b) AgCl(s) Ag + (aq) + Cl (aq) (c) Mg 3 (PO 4 ) 2 (s) 3 Mg 2+ (aq) + 2...
-
What is the pH of 0.10 M NaOH? Strategy We know that NaOH is a strong base, so it will completely dissociate in solution. This gives us [OH ]. We can use the K w relationship between [OH ] and [H 3...
-
Indicate what's wrong with the following arguments: (a) As the firm borrows more and debt becomes risky, both stockholders and bondholders demand higher rates of return. Thus by reducing the debt...
-
Jerrys Window Service received $14,000 from a client on February 20. This payment was an advance payment for 7 months of window cleaning starting March 1. The window cleaning services are provided...
-
Find the Errors 28. Each of the following program segments has some errors. Locate as many as you can. A) Cout < < "Enter a number: "; Cin < < number1; Cout < number1, number2; quotient = double...
-
Specialty Services performs high-end security services. The firm has computed its total direct labor cost for the next quarter to be $111,000. The firm expects two different types of clients during...
-
Volkswagen's Greenwashing Backfires: The Road to Redemption Describe the concept of greenwashing and explain how VW practiced greenwashing and state its implication on the company. Summarize VW's...
-
A project is calculated to have a critical path duration of 10 days with 3 activities on the critical path and corresponding standard deviations A (standard deviation is 2) B (standard deviation is...
-
The maximum recommended speed for a 250-mm-diameter abrasive grinding wheel is 2000 rev/min. Assume that the material is isotropic; use a bore of 20 mm, = 0.24, and a mass density of 3320 kg/m3, and...
-
Will the prediction interval always be wider than the estimation interval for the same value of the independent variable? Briefly explain.
-
Data taken from a stressstrain test for a ceramic are given in the table. The curve is linear between the origin and the first point. Plot the diagram, and determine the modulus of elasticity and the...
-
Data taken from a stressstrain test for a ceramic are given in the table. The curve is linear between the origin and the first point. Plot the diagram, and determine approximately the modulus of...
-
The stressstrain diagram for a steel alloy having an original diameter of 0.5 in. and a gage length of 2 in. is given in the figure. Determine approximately the modulus of elasticity for the...
-
Walsh and Abraham began a partnership by investing 5 3 2 0 , 0 0 0 and $ 4 0 0 , 0 0 0 , respectively. They agreed to share net income / losses by allowing a 1 0 % interest allowance on their...
-
If a company has net income of $ 8 , 5 0 0 , 0 0 0 , average share of common stock outstanding of 2 , 0 0 0 , 0 0 0 , average total stockholders' equity of $ 1 5 4 , 4 0 0 , 0 0 0 and annual...
-
On January 1, 2022, Broker Corporation issued $2,400,000 par value 11% coupon rate, 11-year bonds which pay interest each December 31. If the market rate of interest was 13%, what was the issue price...

Study smarter with the SolutionInn App


