A compound with the formula C 5 H 8 could have either of the following structures. If
Question:
A compound with the formula C5H8 could have either of the following structures. If the addition of an equimolar amount of molecular hydrogen yields only a single pure compound, which structure is likely to be correct?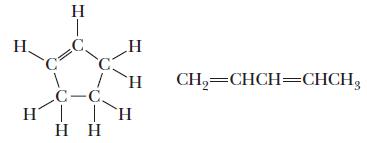
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The structure on the left is likely to be correctThis is because the structure on th...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Create an accompanying report that draws on relevant theories and concepts within Block 1 to explain and justify the decisions made in producing your team's piece of marketing communication. Context...
-
Although the death penalty in India has similarities to the United States, the Indian Supreme Court and the Presidents of India have successfully halted nearly all executions. How have t he Supreme...
-
Perform the same computation as Sec. 24.1, but compute the amount of heat requited to raise the temperature of 1200 g of the material from -150 to 100C. Use Simpsons rule for your computation, with...
-
The inductor of Problem 1.13 is to be operated from a 60-Hz voltage source. (a) Assuming negligible coil resistance, calculate the rms inductor voltage corresponding to a peak core flux density of...
-
A thin electrical heater dissipating 4000 W/m 2 is sandwiched between two 25-mm-thick plates whose exposed surfaces experience convection with a fluid for which Too = 20C and h = 400 W/m 2 . K. The...
-
The \(\frac{1}{2}\)-in.-diameter hose shown in Fig. P8.68 can withstand a maximum pressure of \(200 \mathrm{psi}\) without rupturing. Determine the maximum length, \(\ell\), allowed if the friction...
-
In early 2010, while reviewing Huffman Inc.s 2009 financial records, the accountant discovered several errors. For each of the following errors, indicate the effect on net income (i.e.,...
-
A zookeeper finds an escaped monkey hanging from a light pole. Aiming her tranquilizer gun at the monkey, she kneels 10.0 m above the ground. At the same moment that the monkey drops a banana, the...
-
Suppose the Scope Triangle were modified as follows: Resource Availability occupies the center, and the three sides are Scope, Cost, and Schedule. Interpret this triangle as if it were a system in...
-
Describe the most important intermolecular force between molecules of (a) 2-methyl-2-propanol. (b) 2-butanone.
-
The temperature T'in a freezer is always within 5F of -10F. Write an absolute value inequality that describes possible temperatures in the freezer. Give the temperature range of T.
-
Assume that a business collects two-thirds of its sales revenue in the month of sale and the remaining one-third in the following month. Required: a How much revenue has the business actually earned...
-
The Speedy Answering Service Business was started on 1 July of the current year to answer the phones of doctors, lawyers and accountants when they are away from their offices. The following...
-
The Gomez Folding Chair Company has 400 chairs (at \(\$ 15\) each) in its beginning inventory for July. It makes the following purchases and sales of chairs during July: Required: Calculate the cost...
-
The Costello Tax Services Business was established on 1 March of the current year to help clients with their tax planning. During January, the business entered into the following transactions....
-
When Janet Chiu arrived at her shop on the morning of 15 June 20X3, she found that thieves had broken in overnight and stolen much of her merchandise. The agent of Alright Insurance Company agreed to...
-
Carter wants to retire from his florist business, and his long-time employee, Howard, would like to take over the business. Identify the tax issue(s) posed by the facts presented. Determine the...
-
Inexhaustible collections of ONPOs are not required to be capitalized or depreciated, if certain criteria are met. Why is this so, and what accounting and reporting recognition, if any, is required...
-
When magnesium is placed in an acid solution, hydrogen gas is given off. Is magnesium or hydrogen the better reducing agent?
-
A displacement reaction is an oxidation-reduction reaction in which one element displaces another from solution. In each of the following displacement reactions identify the element that is oxidized...
-
Hydrogen sulfide gas dissolves in water and dissociates very slightly: H 2 S 2H + + S 2 -. How would the acidity of the solution be affected by a. Increasing the pressure of H 2 S? b. Raising the...
-
ABC Firm issued a 10 year bond semi-annual bond with an annual rate of 4% some seven years ago. What is the price of the bond now (three years to go) if the annual rate of interest demanded by...
-
Why is the study of financial business theories including budgets is important overall Program of Study in business? For example, if you want to pursue a major in Marketing- why is the study of...
-
Should Bitcoin and other cryptocurrency be regulated and if yes in what ways can they be regulated ?

Study smarter with the SolutionInn App


