A displacement reaction is an oxidation-reduction reaction in which one element displaces another from solution. In each
Question:
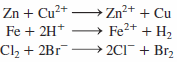
Transcribed Image Text:
Zn + Cu2+ Fe + 2H* Cl, + 2Br- Zn²+ + Cu Fe2+ + H2 2CI+ Br, 111
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (10 reviews)
Oxidized ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A disproportionation reaction is an oxidation-reduction reaction in which the same substance is oxidized and reduced. Complete and balance the following disproportionation reactions:
-
Complete and balance the following half-reactions. In each case indicate whether the half-reaction is an oxidation or a reduction.
-
Complete and balance the following half-reactions. In each case indicate whether the half-reaction is an oxidation or a reduction. Discuss.
-
You have been asked by the principal investigator of a qualitative research project to suggest ideas for maintaining confidentiality of private, sensitive information obtained from human...
-
Halle describes "Third-worldism" as an automatic resistance to change among poorer countries to proposals that come from richer countries. What could be done to overcome this distrust?
-
Al Medina, D.D.S., opened an incorporated dental practice on January 1, 2017. During the first month of operations, the following transactions occurred. 1. Performed services for patients who had...
-
Using the AISC equations, select from Appendix B the lightest-weight wide-flange A992 steel column that is \(12 \mathrm{ft}\) long and supports an axial load of 20 kip. The ends are pinned.
-
The following selected transactions were completed by Niles Co. during March of the current year: Mar. 1. Purchased merchandise from Haas Co., $43,250, terms FOB shipping point, 2/10, n/eom. Prepaid...
-
Research and explore the cutting-edge energy below. Zero-point Energy Helpful links: https://www.scientificamerican.com/article/follow-up-what-is-the-zer/ http://www.calphysics.org/zpe.html...
-
The city manager of University City is finalizing the budget proposal that must be submitted to the city council 60 days prior to the July 1 start of the next fiscal year, FY 20X2. An economic...
-
When magnesium is placed in an acid solution, hydrogen gas is given off. Is magnesium or hydrogen the better reducing agent?
-
Hydrogen sulfide gas dissolves in water and dissociates very slightly: H 2 S 2H + + S 2 -. How would the acidity of the solution be affected by a. Increasing the pressure of H 2 S? b. Raising the...
-
To which of the following tax-exempt organizations may the UBIT apply? a. Red Cross. b. Salvation Army. c. United Fund. d. College of William and Mary. e. Rainbow, Inc., a private foundation. f....
-
Q.2. For a point-to-point link of length 30 km, at what value of link transmission rate (in Mbps or Megabits-per-sec) would the link propagation delay (at a speed of 2 108 m/sec) equal the...
-
Create C++ programs to replicate these two UNIX(UBUNTU) commands. TYPE generates output and prints it to standard out and CAT that processes information given to it on standard input: TYPE CAT
-
15 Following are some transactions and events of Business Solutions. February 26 The company paid cash to Lyn Addie for eight days' work at $120 per day. March 25 The company sold merchandise with a...
-
How to store text file data into a dictionary in JavaScript? I have having an issue with that part. In the text file, there's an english term and the spanish translations of the word. The dictionary...
-
what is one of the disadvantages of incorporating continual improvement as a facility management goal.
-
Using the information given in Problem 1.1 and the properties of the standard atmosphere given in Appendix G, estimate the number of molecules per cubic inch at an altitude of 250,000 ft. Data From...
-
4. Jobe dy -Y 2 et by
-
A vessel containing a liquid is opened inside an evacuated chamber. Will you see a liquidgas interface if the volume of the initially evacuated chamber is a. Less than the critical volume, b. A...
-
Calculate S R for the reaction H 2 (g) + Cl 2 (g) 2HCl (g) at 870. K. Omit terms in the temperature dependent heat capacities higher than T 2 /K 2 .
-
Use the result of Problem P3.10 to derive a formula for (CV /V ) T for a gas that obeys the RedlichKwong equation of state, RT 1 a Vm - b VT VVm + b)' T VVm P:
-
1. Research existing case law to analyze how the Supreme Court of the United States has ruled in an effort to achieve the Sixth Amendment's chief goal. a. Analyze at least four cases (three of which...
-
Jason is 25 years old and has a new job in Web development. He wants to make sure he is financially sound when he retires 30 year later. He plans to invest the same amount to a retirement account for...
-
Henry, a single taxpayer with a marginal tax rate of 35 percent (taxable income is $300,000 before considering any of the items below), sold all of the following assets during the year: Holding...

Study smarter with the SolutionInn App


