For each of the following reactions, identify the BrnstedLowry acids and bases. What are the conjugate acid/base
Question:
For each of the following reactions, identify the Brønsted–Lowry acids and bases. What are the conjugate acid/base pairs?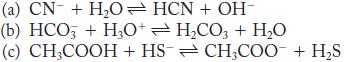
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a CN base HCNacid and H2O...View the full answer

Answered By

Nandkumar Ghadge
From my childhood I was fascinated by this world. Knowledge is scattered around that you have to garnish and distribute among others and is to be used for welfare of mankind.
I have 4 years of teaching experience to undergraduate students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Write an equation for each of the following equilibria, and use Table 3.1 to identify the pK a value associated with the acidic species in each equilibrium. (a) Ammonia acting as a base toward the...
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
Using the pK a values in Table 3.1, calculate the equilibrium constant for each of the following reactions. (a) NH 3 acting as a base toward the acid HCN (b) F acting as a base toward the acid HCN...
-
Determine whether the given functions are even, odd, or neither. a. x sin(x) c. x cos(x) e. x sin(x) + x sin(x) f. x sin(x) + x cos(x) even h. x sin(x) + x cos(x) j. x cos (x) + x cos(x) b. x sin(x)...
-
Consider the heat exchanger of Problem 11.14. After several years of operation, it is observed that the outlet temperature of the cold water reaches only 95C rather than the desired 120C for the same...
-
Consider a cogeneration power plant that is modified with reheat and that produces 3 MW of power and supplies 7 MW of process heat. Steam enters the high-pressure turbine at 8 MPa and 500°C and...
-
Marijuana Use The percent of college students marijuana use for a sample of 95,761 students is shown in the pie chart. Find the probability of each event. (a) Randomly selecting a student who never...
-
The two mooring cables exert forces on the stern of a ship as shown. Represent each force as a Cartesian vector and determine the magnitude and direction of theresultant. F,= 150 lb = 200 lb 50 ft...
-
At least two economic trends which are impacting the H&M ' s operations ( e . g . , could be trends in demand and supply of their products / services , unemployment rate, consumer confidence in the...
-
Write the formula, and give the name of the conjugate acid of each of the following bases.
-
Write the formula and name for the conjugate acid of the following substances.
-
Williams Corporation imports, from a number of German manufacturers, large machining equipment used in the tooling industry. On June 1, the company received delivery of a piece of machinery with a...
-
How will you stay informed about new digital communication technologies in the future? Do you think you or institutions for which you work should be responsible for keeping you informed about them...
-
Take another look at figure 16.2, the heuristic for structuring content visually. In your opinion, which of the three phases will be the most difficult for students whose educational background has...
-
Working with another student, find technical communication job ads that interest each of you. Then take turns interviewing each other for the job you chose. As the interviewer, plan interview...
-
You probably have learned a lot of research skills in school. You may also have done client-based projects in some of your technical communication courses, where you had to apply these skills to the...
-
Susan, the technical communication manager at a tax and accounting software company, said, "I want writers to become experts on the product they document." Assuming you do not have such expertise...
-
Software Solution (SOS) helps subscribers solve software problems. All transactions are made over the telephone. For the year 2009, 10 engineers, most of whom are recent graduates, handled 100,000...
-
APC16550D UART has a clock running at18.432 MHz and its baud rate is set to 2000.Determine the HEX contents of its DLM and DLL registers. Please can you explain step by step and in detail how you get...
-
An electric welding machine uses a current of 400 A. The machine has a transformer whose primary coil has 400 turns and draws 4 A from a 240-V power line. How many turns does the secondary coil have?...
-
The primary coil of a transformer has 100 turns and its secondary coil has 400 turns. Ignoring the resistance of the coils, compare the input power to the primary coil with the output power from the...
-
Given a coil of wire and a small lightbulb, how can you tell whether the current in another coil is direct or alternating without touching the second coil or its connecting wires?
-
What is the primary difference between secured and unsecured loans? Payton needs money today, so he decides to take out a loan for $627 with a payday lender. The lender expects the money back in 5...
-
1. Why are Us Treasury securities are referred to as Risk free rate of return? 2. Know what the slope of yeild curve can tell us about the economy in the future 3. what the slope of yeild can tell us...
-
Explain how a business finances its operations and expansion. CURRENT RATIO (Current Assets / Current Liabilities) TOTAL ASSET TURNOVER RATIO (Total Revenue / Total Assets) Current Assets 53.7 Total...

Study smarter with the SolutionInn App


