In the process of isolating iron from its ores, carbon monoxide reacts with iron(III) oxide, as described
Question:
In the process of isolating iron from its ores, carbon monoxide reacts with iron(III) oxide, as described by the following equation: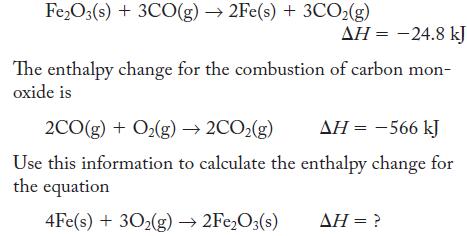
Transcribed Image Text:
Fe2O3(s) + 3CO(g) → 2Fe(s) + 3CO₂(g) ΔΗ = -24.8 kJ The enthalpy change for the combustion of carbon mon- oxide is 2CO(g) + O₂(g) → 2CO₂(g) AH = -566 kJ Use this information to calculate the enthalpy change for the equation 4Fe(s) + 302(g) → 2Fe₂O3(s) AH = ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The balanced chemical equation for the reaction of carbon monoxide CO ...View the full answer

Answered By

Mohiddin Shaik
I have Bachelors degree in Electrical and Electrons engineering
Masters degree in Control systems
I have 3 years experience in Teaching
I have 5 years years experience as Q&A expert.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
What does an energy-level diagram for the reverse reaction from Exercise 5.72 look like? Exercise 5.72 In the process of isolating iron from its ores, carbon monoxide reacts with iron(III) oxide, as...
-
Draw an energy-level diagram that represents the Hesss law calculation in Exercise 5.72. Exercise 5.72 In the process of isolating iron from its ores, carbon monoxide reacts with iron(III) oxide, as...
-
Solid oxide fuel cells (SOFC) have been proposed as an alternative energy technology for use in large stationary power applications (1 to 10MWof electrical power). These devices have an ion...
-
How many gallons of mercury (sg = 13.54) would weigh the same as 5 gal of castor oil, which has a specific weight of 59.69 lb/ft 3 ?
-
Repeat Example 16.8, except that the ore contains 3 wt% Cu 2 O
-
If an even function (x) has a local maximum value at x = c, can anything be said about the value of at x = -c? Give reasons for your answer.
-
Thomas and James, who were operating separate competing businesses, decided on 1 July 2025 to form a partnership by contributing cash, assets and liabilities of their respective businesses. At that...
-
The following information was taken from Lamberson Companys accounting records: Additional information for the year: a. b. Dividends declared and paid totaled $700. c. On January 1,2016, convertible...
-
When an object is placed at the focus of a concave mirror, the image formed is (i) at infinity (ii) real and inverted, and (iii) highly magnified (or highly enlarged) M E A D C B P F At infinity N
-
Chumpy Lighting Limited manufactures a wide variety of light bulbs which it sells to lighting shops and builders merchants through wholesale distributors. It also sells direct to the big UK...
-
Given the thermochemical equations 2Cu(s) + Cl(g) 2CuCl(s) 2CuCl(s) + Cl(g) 2CuCl2(s) find the enthalpy change for Cu(s) + Cl(g) CuCl(s) AH = -274.4 kJ AH = -165.8 kJ AH = ?
-
Calculate H for the reaction Zn(s) + --0(g) ZnO(s) 2 given the equations AH = ? Zn(s) + 2HC1(aq) ZnCl (aq) + H(g) 2H(g) + O(g) 2HO(l) = -152.4 kJ ZnO(s) + 2HC1(aq) ZnCl(aq) + HO(0) AH = -90.2 kJ...
-
After decades of falling domestic oil production, by 2014-2015 output was surging in the United States. The reason? Technological changes in producing crude oil and natural gas from shale (hydraulic...
-
A closed vessel of \(0.15 \mathrm{~m}^{3}\) capacity contains a mixture of methane \(\left(\mathrm{CH}_{4} ight)\) and air, the air being \(20 \%\) in excess of that required for chemically correct...
-
A gas injection system supplies a mixture of propane \(\left(\mathrm{C}_{3} \mathrm{H}_{8} ight)\) and air to a spark-ignition engine, in the ratio of volumes of 1:30. The mixture is trapped at \(1...
-
How can the equation of state in the form of a relationship between pressure, volume and temperature be used to extend limited data on the entropy of a substance. A certain gas, A, has the equation...
-
A mixture of one part by volume of vapourised benzene to 50 parts by volume of air is ignited in a cylinder and adiabatic combustion ensues at constant volume. If the initial pressure and temperature...
-
A mixture containing equal volumes of carbon dioxide \(\left(\mathrm{CO}_{2} ight)\) and hydrogen \(\left(\mathrm{H}_{2} ight)\) is contained in a rigid vessel. It is ignited at \(60^{\circ}...
-
Look one more time at Table. a. Suppose you knew the bond prices but not the spot interest rates. Explain how you would calculate the spot rates. (You have four unknown spot rates, so you need four...
-
The Taylor's series expansion for cosx about x = 0 is given by: where x is in radians. Write a user-defined function that determines cosx using Taylor's series expansion. For function name and...
-
How do you expect S m for an ion in solution to change as the charge increases at constant ionic radius?
-
It takes considerable energy to dissociate NaCl in the gas phase. Why does this process occur spontaneously in an aqueous solution? Why does it not occur spontaneously in CCl 4 ?
-
Calculate S R for the reaction Ba(NO 3 ) 2 (aq) + 2KCl(aq) BaCl 2 (s) + 2KNO 3 (aq).
-
what ways do chromosomal abnormalities, such as chromosomal mosaicism, ring chromosomes, and marker chromosomes, challenge our understanding of chromosomal biology, genetic diagnosis, and...
-
For growing numbers of students, college is not just time of learning, partying and growing into young adulthood; it is fast becoming a place for building a business. More than 3,000 colleges and...
-
Assume that you are a consultant to assess the potential for the location decision. Please provide your answer to the following questions: 1. What are the two key criteria for Amazon to choose the...

Study smarter with the SolutionInn App


